Timekeepers of Immunity: Dissecting the BMAL1-Dependent Anti-Inflammatory Action of REV-ERBα Agonist SR9009
Abstract
The circadian clock plays a pivotal role in regulating immune responses, yet the interplay between its core components in inflammatory control remains incompletely understood. This blog explores findings from a study investigating the REV-ERBα agonist SR9009 in the context of macrophage-driven inflammation. The research demonstrates that SR9009 effectively suppresses IL-1β and IL-18 production by inhibiting the NLRP3 inflammasome pathway, a process shown to be critically dependent on the expression of BMAL1. While SR9009 reduces systemic inflammation in LPS-induced endotoxemia, its metabolic effects—especially in BMAL1-deficient mice—reveal a paradox: energy conservation is impaired during acute stress. These insights underscore the complexity of targeting circadian regulators for anti-inflammatory therapy. The study highlights the potential of clock-based pharmacology while cautioning against unintended systemic consequences, pointing toward more nuanced, time-aware strategies for treating inflammatory diseases.
Circadian Clocks and Immunity: A New Frontier in Inflammation Control
The circadian clock does far more than regulate sleep–wake cycles—it orchestrates a vast array of physiological processes, including metabolism, hormone secretion, and immune function. At the core of this system lies a tightly regulated transcriptional feedback loop, prominently featuring the genes BMAL1 and REV-ERBα. These transcription factors operate in opposition, driving rhythmic gene expression across tissues to align internal biological activity with the external environment.
In recent years, research has illuminated a critical connection between circadian regulation and the innate immune response. Macrophages, the first responders in host defense, exhibit time-of-day-dependent fluctuations in inflammatory activity. They detect microbial components like lipopolysaccharide (LPS) and respond by producing cytokines such as interleukin-1β (IL-1β), interleukin-18 (IL-18), and tumor necrosis factor-alpha (TNFα). These responses are not static—they oscillate with the circadian cycle, influenced by clock-controlled gene expression. Disruption of this rhythmicity, whether due to environmental stress or genetic mutation, has been linked to heightened inflammatory states and increased vulnerability to disease.
Among the circadian regulators, REV-ERBα has emerged as a compelling target for modulating immune activity. As a nuclear receptor and transcriptional repressor of BMAL1, REV-ERBα plays a pivotal role in maintaining circadian balance. Notably, pharmacological activation of REV-ERBα using synthetic ligands such as SR9009 has demonstrated anti-inflammatory effects in preclinical models. Yet, a fundamental question remains: does REV-ERBα act independently to suppress inflammation, or is its efficacy contingent on the presence of BMAL1?
A recent study by Hong et al. (2021) provides new insights into this relationship. By examining the effects of SR9009 in both wild-type and BMAL1-deficient macrophages, the researchers explored how circadian regulators intersect with the NLRP3 inflammasome pathway, a key mechanism in IL-1β and IL-18 maturation and secretion. Their findings shed light on the nuanced interplay between circadian transcription factors and innate immunity—an area of growing interest for the development of chronotherapeutic strategies in inflammatory disease.
BMAL1 and REV-ERBα: Dual Timekeepers in the Inflammatory Cascade
The molecular circadian clock is governed by a transcriptional–translational feedback loop, with BMAL1 (Brain and Muscle ARNT-Like 1) and CLOCK forming a heterodimer that activates the expression of several downstream clock genes, including Period (Per), Cryptochrome (Cry), and Rev-erbα (NR1D1). These in turn feed back to repress BMAL1/CLOCK activity, generating a ~24-hour cycle in gene expression that tightly regulates cellular and systemic homeostasis.
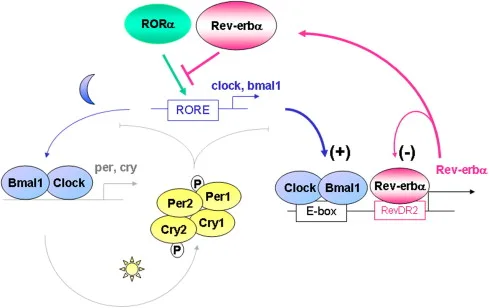
In immune cells, this circadian machinery has profound functional implications. Macrophages, for example, exhibit rhythmic patterns in phagocytic capacity, antigen presentation, and cytokine production. The transcriptional landscape of these cells is dynamically reshaped across the day, influencing how they respond to pathogens or damage signals. Among the most compelling insights in recent years is the discovery that core clock genes not only modulate immune function but also serve as direct repressors or enhancers of inflammatory signaling pathways.
BMAL1 has been shown to exert anti-inflammatory effects by inhibiting pathways such as NF-κB and HIF-1α, and its deletion in myeloid cells leads to heightened LPS sensitivity and worsened outcomes in models of sepsis. Conversely, REV-ERBα, traditionally viewed as a repressor of BMAL1 transcription, also plays a protective role against inflammation by directly suppressing pro-inflammatory mediators, including IL-6 and components of the NLRP3 inflammasome. The latter is particularly significant, as the NLRP3 inflammasome is a central hub in macrophage-driven inflammation, controlling the maturation and release of IL-1β and IL-18 in response to microbial stimuli like LPS.
These dual roles place BMAL1 and REV-ERBα in a unique position: both act as negative regulators of inflammation, but through distinct and possibly overlapping mechanisms. The synthetic ligand SR9009, an agonist of REV-ERBα, offers a tool to pharmacologically interrogate these interactions. Previous work has shown that SR9009 can attenuate inflammation and metabolic dysfunction in models of colitis and neuroinflammation. However, whether its efficacy depends on the functional presence of BMAL1 has remained unresolved—until now.
In the study by Hong et al., this question was addressed using BMAL1-floxed mice with myeloid-specific knockout, alongside wild-type controls, to isolate the contribution of BMAL1 in REV-ERBα-driven anti-inflammatory responses. The results provide a deeper mechanistic understanding of how circadian components intersect in regulating the NLRP3–IL-1β axis, offering new perspectives for timing and targeting inflammation therapeutically.
Disarming the Inflammasome: How SR9009 and BMAL1 Coordinate IL-1β Suppression
To explore the interplay between REV-ERBα activation and BMAL1 in regulating inflammation, Hong et al. conducted a series of in vitro and in vivo experiments centered around LPS-induced inflammatory responses in macrophages. Their model system included both wild-type bone marrow-derived macrophages (BMDMs) and BMDMs from BMAL1-deficient mice (Bmal1^f/f;LysM^cre/+), enabling a precise dissection of gene-specific contributions.
Time-of-Day Dependency in Cytokine Response
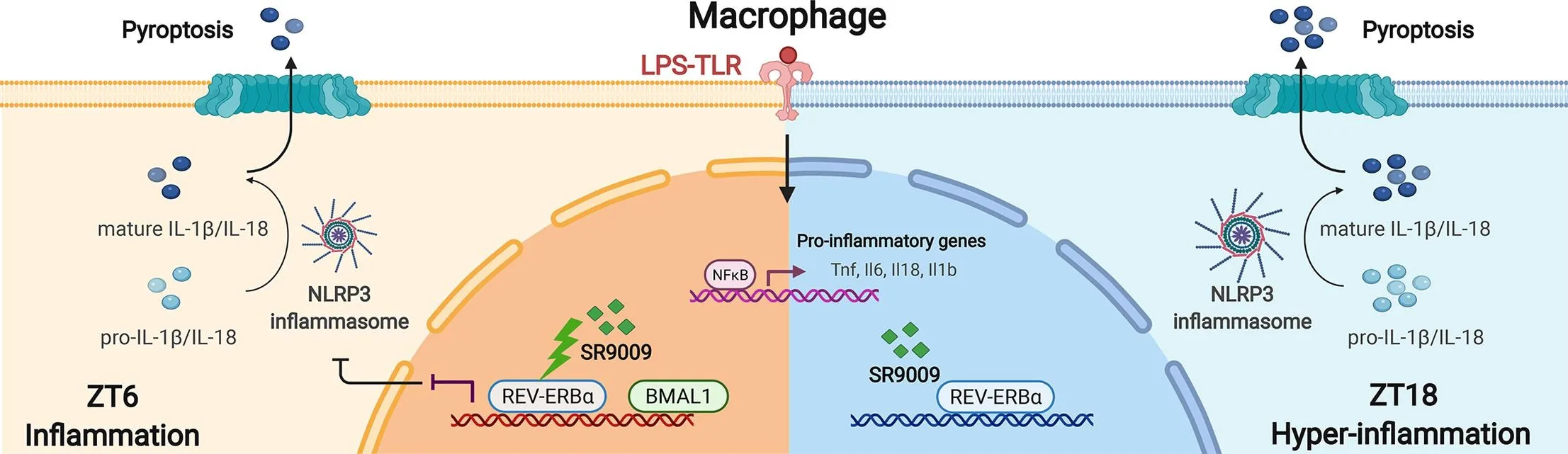
The researchers began by confirming that LPS-induced inflammation in macrophages follows a circadian pattern. When BMDMs were challenged with LPS at different circadian times—defined by Zeitgeber Time (ZT)—they observed a significantly higher expression of pro-inflammatory cytokines (Tnf, Il6, Il1b, Il18) at ZT18 compared to ZT6. This confirmed that macrophage inflammatory responses are time-gated, with greater cytokine output occurring during the late active/resting phase, consistent with known oscillations in BMAL1 and REV-ERBα expression.
SR9009 Suppresses Inflammation—But Not Uniformly
Administration of SR9009, a selective REV-ERBα agonist, significantly reduced mRNA levels of Tnf, Il6, Il1b, and Il18 in LPS-treated macrophages at both ZT6 and ZT18. Interestingly, while the suppression of IL-6 was slightly stronger at ZT18, SR9009 was broadly effective across both time points, suggesting its anti-inflammatory effect is largely independent of circadian timing—at least at the transcriptional level.
However, a more nuanced picture emerged at the protein level. In wild-type macrophages, SR9009 treatment markedly reduced both the pro-form and mature form of IL-1β and IL-18, key cytokines processed via the NLRP3 inflammasome. In BMAL1-deficient macrophages, however, SR9009 failed to suppress the protein levels of these cytokines, despite reducing their mRNA transcripts. This disconnect pointed toward a post-transcriptional mechanism—namely, inflammasome activation—that requires BMAL1.
BMAL1 Is Required for NLRP3 Repression
The researchers then turned their focus to the NLRP3 inflammasome, the molecular complex responsible for cleaving pro-IL-1β and pro-IL-18 into their active forms. In wild-type BMDMs, SR9009 suppressed Nlrp3 gene expression and protein levels following LPS stimulation. This repression was consistent with prior studies showing that REV-ERBα can bind the Nlrp3 promoter and inhibit transcription.
In contrast, BMAL1-deficient macrophages showed elevated NLRP3 expression, and SR9009 failed to repress NLRP3 at either the mRNA or protein level—especially at ZT18, when BMAL1 expression is naturally lower. These findings underscore that BMAL1 is indispensable for REV-ERBα to exert full control over inflammasome activation. Without BMAL1, SR9009’s effects are limited to upstream transcriptional modulation but cannot disrupt the final step of cytokine maturation.
In Vivo Confirmation: The Endotoxemia Model
To validate these results in a physiological setting, the study employed a mouse model of endotoxemia, induced by intraperitoneal injection of LPS. Mice were pre-treated with SR9009 prior to LPS challenge, and peritoneal macrophage populations were analyzed via flow cytometry.
In wild-type mice, SR9009 reduced the recruitment of inflammatory monocytes (small peritoneal macrophages, SPMs) and suppressed IL-1β production in both resident (LPMs) and infiltrating (SPMs) macrophages. However, in BMAL1-deficient mice, SR9009 failed to suppress IL-1β in SPMs, though some reduction in TNFα and systemic IL-1β levels was still observed. This again confirmed that BMAL1 is essential for SR9009’s full anti-inflammatory effect on inflammasome-driven cytokines, particularly IL-1β and IL-18.
A Metabolic Trade-Off: When Anti-Inflammatory Therapy Impairs Energy Recovery
While SR9009 demonstrated potent anti-inflammatory effects through REV-ERBα activation, its influence extended beyond immune signaling into systemic energy metabolism—a crucial aspect often altered during acute inflammation. In response to LPS-induced endotoxemia, mammals typically enter a hypometabolic state, characterized by reduced oxygen consumption (VO₂), carbon dioxide output (VCO₂), food intake, and overall energy expenditure. This shift is thought to be an adaptive mechanism—a means to conserve energy during an immune crisis.
In their in vivo experiments, Hong et al. used metabolic phenotyping to track these physiological changes in both wild-type and BMAL1-deficient mice. Following LPS administration, all mice exhibited the expected hypometabolic response. However, the magnitude and duration of this state were significantly greater in BMAL1-deficient animals, indicating a heightened susceptibility to energy collapse during systemic inflammation.
Interestingly, pre-treatment with SR9009 exacerbated the hypometabolic state in BMAL1-deficient mice, further reducing VO₂, VCO₂, and energy expenditure beyond that observed in vehicle-treated controls. These effects were accompanied by a delay in recovery from the LPS-induced metabolic downturn and a greater reduction in food intake, which served as an additional indicator of prolonged sickness behavior.
At the cellular level, this observation was supported by mitochondrial respiration assays. In BMDMs, SR9009 treatment did not restore mitochondrial function in BMAL1-deficient cells. Instead, these macrophages displayed a significant decrease in ATP-linked respiration and spare respiratory capacity, underscoring their diminished ability to meet energy demands under stress. This impairment may stem from an inability to engage oxidative phosphorylation (OxPhos) effectively during immune activation.
Taken together, these results suggest that while REV-ERBα activation dampens inflammation, it also modulates energy balance in a BMAL1-dependent manner. In the absence of BMAL1, SR9009 may tip the metabolic scale unfavorably—heightening energy suppression when conservation is already critical. This paradox highlights a potential trade-off: targeting inflammation via circadian pathways could inadvertently compromise host metabolic resilience if the underlying clock machinery is disrupted.
These findings are especially relevant in conditions like sepsis, where the interplay between immune response and metabolic stability can determine survival outcomes. They also raise important considerations for the therapeutic use of REV-ERBα agonists, particularly in patients with circadian misalignment or underlying metabolic disorders.
Synchronizing Immunity: Clock-Based Strategies for Future Anti-inflammatory Therapies
The work by Hong et al. offers compelling evidence that circadian clock components are not merely passive timekeepers but active regulators of immune homeostasis. Through the pharmacological activation of REV-ERBα with SR9009, the study demonstrates a powerful suppression of IL-1β and IL-18 production in macrophages, mediated via inhibition of the NLRP3 inflammasome. Crucially, this anti-inflammatory effect is shown to be BMAL1-dependent at the post-transcriptional level, highlighting an underappreciated interdependence between these two core circadian regulators.
From a therapeutic standpoint, these findings open new avenues for clock-based intervention in inflammatory diseases, particularly those involving aberrant IL-1β signaling such as sepsis, gout, and autoimmune conditions. By targeting REV-ERBα, it may be possible to tune down pathological inflammation without broadly suppressing immune function.
However, the study also underscores a critical caveat: the metabolic consequences of REV-ERBα activation are not uniformly beneficial. In the absence of BMAL1, SR9009 exacerbated hypometabolic states and delayed recovery from acute inflammatory stress, suggesting that chronotherapeutic strategies must account for the broader systemic roles of clock genes. For instance, BMAL1 not only supports cytokine repression but also helps maintain mitochondrial function and energy balance during immune activation.
Future research should aim to:
- Decipher tissue-specific roles of BMAL1 and REV-ERBα in immune-metabolic regulation.
- Explore the temporal dynamics of inflammasome activation in relation to circadian gene expression.
- Investigate combinatorial therapies that pair clock-targeting agents like SR9009 with metabolic stabilizers.
- Translate these findings into human models, particularly in shift workers, patients with circadian misalignment, or those at risk of inflammatory disease.
Ultimately, this study reinforces the notion that inflammation and metabolism are deeply interwoven with circadian biology. Precision therapies that respect and leverage this temporal framework may offer a new frontier in treating complex inflammatory disorders—not by shutting down the immune system, but by synchronizing it.
 LinkPeptide
LinkPeptide