Pegcetacoplan: A Breakthrough in Complement-Mediated Disease Therapy and Its Expanding Clinical Impact
Abstract
Pegcetacoplan is a PEGylated C3 inhibitor that represents a significant advancement in the treatment of complement-mediated diseases. By targeting complement component 3 (C3) and its fragment C3b, Pegcetacoplan regulates both extravascular and intravascular hemolysis, offering a novel approach to conditions such as paroxysmal nocturnal hemoglobinuria (PNH), C3 glomerulopathy, and autoimmune hemolytic anemia. Clinical trials have demonstrated its superiority over existing C5 inhibitors, particularly in improving hemoglobin levels and reducing the need for transfusions in PNH patients. The pharmacokinetic and pharmacodynamic properties of Pegcetacoplan, alongside its efficacy in multiple therapeutic areas, highlight its potential as a versatile treatment option. Ongoing research continues to explore its application in additional complement-mediated disorders, making Pegcetacoplan a key therapeutic agent with broad implications for future clinical practice.
Pegcetacoplan: A New Era in Complement-Mediated Disease Therapy
Paroxysmal nocturnal hemoglobinuria (PNH) is a rare, acquired, clonal, nonmalignant hematologic disease characterized by complement-mediated hemolysis (with or without hemoglobinuria), fatigue, increased susceptibility to thrombosis, and some degree of bone marrow dysfunction. PNH results from the expansion of abnormal hematopoietic clones that lack cell-surface complement inhibitory proteins attached to the membrane through glycosylphosphatidylinositol anchors. At least two of the missing glycosylphosphatidylinositol-linked proteins, CD55 and CD59, are key regulators of the complement pathway and protect host cells from complement-mediated removal. This complement dysregulation leads to chronic hemolysis and thrombosis characteristic of PNH.1,9 Manifestations of hemolysis are due directly to the increased sensitivity of PNH cells to complement.
Pegcetacoplan represents a significant breakthrough in the treatment of complement-mediated diseases, offering a novel approach to managing these conditions. Complement-mediated diseases, such as paroxysmal nocturnal hemoglobinuria (PNH), autoimmune hemolytic anemia, and C3 glomerulopathy, involve dysregulation of the complement system, leading to the destruction of blood cells and tissue damage. These diseases are often difficult to manage, and current treatments have had limitations in addressing the full scope of complement activation.
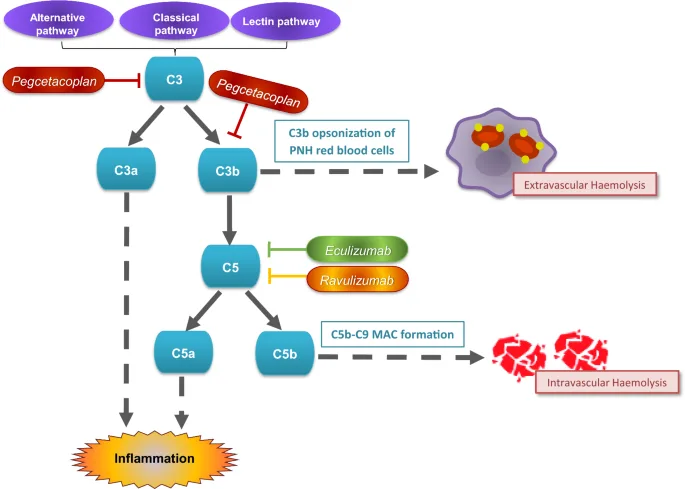
Unlike therapies that target the terminal stages of the complement pathway, Pegcetacoplan specifically inhibits complement component 3 (C3), a central protein in the complement cascade. By binding to C3 and its active fragment C3b, Pegcetacoplan blocks both early and late stages of the complement pathway, providing more comprehensive protection. This dual mechanism prevents both intravascular and extravascular hemolysis, distinguishing Pegcetacoplan from earlier C5 inhibitors, such as eculizumab and ravulizumab, which only block terminal complement activation.
Approved in May 2021 by the U.S. Food and Drug Administration (FDA) for the treatment of PNH, Pegcetacoplan has shown significant clinical benefits. Patients treated with Pegcetacoplan have demonstrated improved hemoglobin levels, reduced need for blood transfusions, and overall better clinical outcomes compared to those using existing therapies. Its innovative mechanism of action and superior efficacy position Pegcetacoplan as a promising treatment for a range of complement-mediated diseases, beyond its current use in PNH.
As research continues to expand, Pegcetacoplan holds promise for treating other conditions linked to complement dysregulation, opening new avenues for therapeutic intervention in rare and complex diseases.
Understanding the Target: How Pegcetacoplan Inhibits Complement C3
Pegcetacoplan’s innovative mechanism of action is central to its success in treating complement-mediated diseases. Unlike other therapies that target the terminal complement pathway, Pegcetacoplan directly inhibits complement component 3 (C3), a critical protein at the heart of the complement system. The complement system is a crucial part of the immune response, playing a role in marking pathogens for destruction, but its dysregulation can lead to damaging effects, such as red blood cell destruction in diseases like paroxysmal nocturnal hemoglobinuria (PNH).
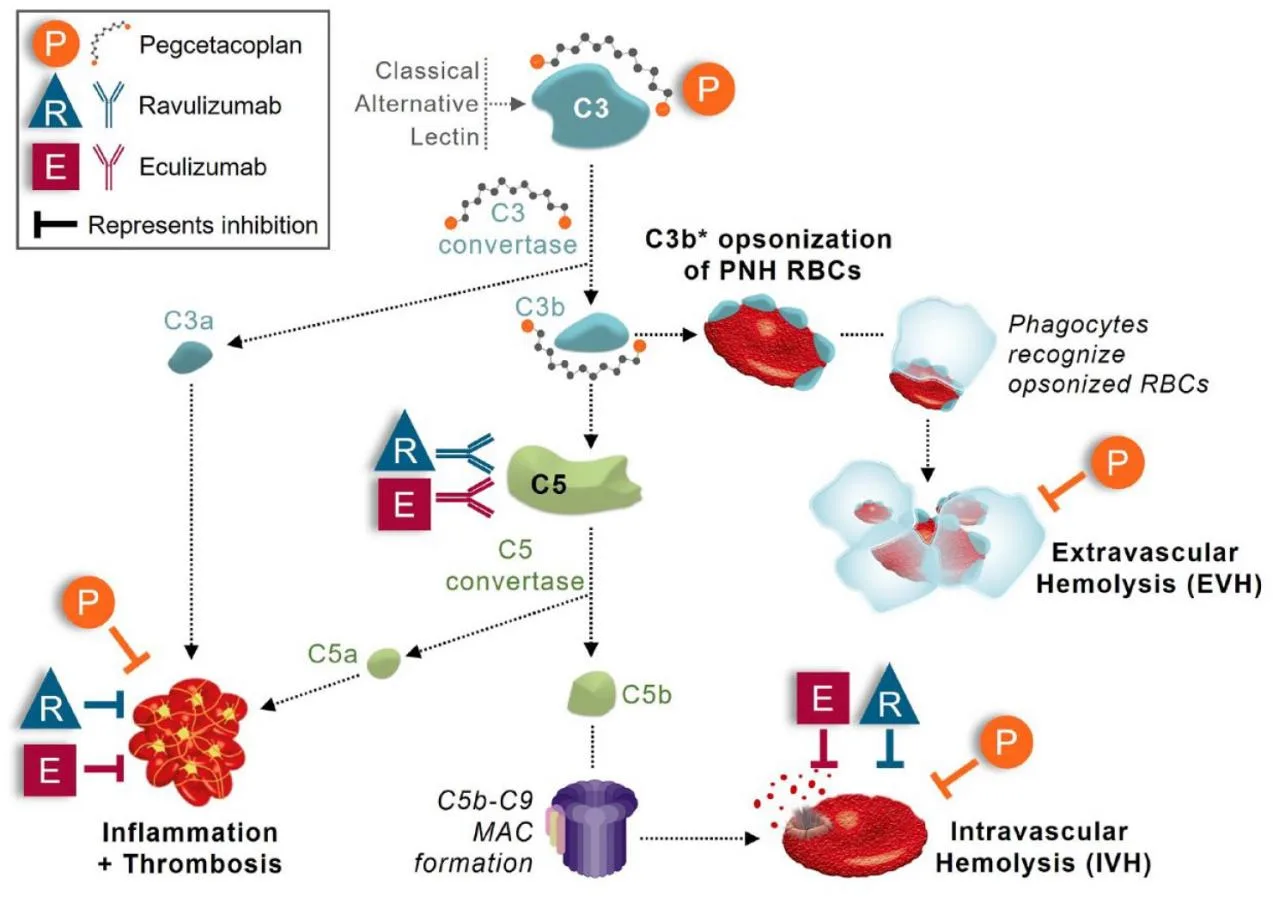
Pegcetacoplan binds to both C3 and its active fragment C3b, preventing their cleavage and blocking the cascade of events that lead to complement activation. By doing so, it inhibits both intravascular hemolysis (the destruction of red blood cells within blood vessels) and extravascular hemolysis (the destruction of red blood cells in organs such as the liver and spleen). This dual action offers a comprehensive approach to preventing red blood cell destruction, which is a key issue in diseases like PNH, where the lack of complement-regulating proteins on the cell surface leaves red blood cells vulnerable to attack.
Traditional therapies, such as eculizumab and ravulizumab, work by inhibiting complement component 5 (C5), which blocks only the terminal stage of the complement pathway. While effective, these therapies do not prevent the earlier stages of complement activation, leaving some patients susceptible to extravascular hemolysis. Pegcetacoplan’s ability to target C3 offers an advantage, as it blocks complement activation earlier in the pathway, addressing both intravascular and extravascular hemolysis. This makes it a more robust treatment option for patients who continue to experience symptoms despite C5 inhibition.
By inhibiting C3, Pegcetacoplan redefines how complement-mediated diseases can be managed, offering broader protection and improving clinical outcomes for patients.
Pegcetacoplan’s Pharmacological Profile and Its Clinical Superiority
Pegcetacoplan’s clinical efficacy is underpinned by its distinct pharmacokinetic and pharmacodynamic properties. As a PEGylated compound, Pegcetacoplan benefits from enhanced stability and prolonged half-life, allowing it to maintain therapeutic levels in the bloodstream for extended periods. In clinical studies, Pegcetacoplan demonstrated dose-proportional exposure over a wide dose range, with peak plasma concentrations reached within 4.5 to 6 days following subcutaneous administration. This predictable pharmacokinetic profile makes it an ideal candidate for long-term therapy, particularly for patients with chronic complement-mediated diseases.
Results from phase I–III clinical trials suggest that pegcetacoplan is efficacious and safe in a broad population of patients with PNH. This has been demonstrated among patients with suboptimal response to prior C5i therapy who achieved superior hemoglobin level improvements with pegcetacoplan compared to eculizumab. In addition, superior hematologic improvements were also achieved with pegcetacoplan versus standard treatment (excluding complement-inhibitors) in complement-inhibitor-naïve patients.
A total of 48 patients were either currently receiving or previously received pegcetacoplan (2019–2023). A total of 12 patients had participated in the PEGASUS clinical trial, continuing treatment after trial completion. Five patients were on combination treatment of C5 inhibition and pegcetacoplan. Mean pegcetacoplan duration was 20.2 months. Indication for pegcetacoplan was EVH on C5 inhibitors (Eculizumab, n = 29, Ravulizumab n = 16, others n = 3) with 35/48 patients requiring blood transfusion within the previous 12 months. Mean hemoglobin and reticulocyte count at pegcetacoplan commencement and after 3 months: 91 g/L and 205 × 109/L and 115.8 g/L and 107 × 109/L, respectively, resulting in mean Hb change of 22.3 g/L. Mean LDH pre- and post-pegcetacoplan was unchanged. Six patients have stopped pegcetacoplan. A total of 32 breakthrough hemolysis (BTH) events occurred in 13/48 patients. A total of 14 events were within clinical trials (reported separately). Six patients experienced 18 acute BTH events outside clinical trials, 7/18 associated with complement activating conditions. Successful clinical management included daily pegcetacoplan subcutaneously for 3 days or single eculizumab doses; these events are manageable with prompt intervention.
Pegcetacoplan presented a favorable safety profile with injection site reactions being the most common TEAE. As expected, rates of injection site reactions decreased as the trials progressed. This finding was consistent with the assumption that these events would become less common as patients gained experience with pegcetacoplan self-administration and were not a barrier to treatment, as numerous QoL gains were reported by patients. No meningococcal infections, a potential concern with complement inhibition, have been observed during clinical studies with pegcetacoplan, indicating that mitigation plans to prevent these infections (i.e. vaccination strategy and prophylactic antibiotic treatment) are effective.
Expanding Horizons: Pegcetacoplan in Emerging Therapeutic Areas
Pegcetacoplan’s success in treating paroxysmal nocturnal hemoglobinuria (PNH) has catalyzed further research into its potential applications in other complement-mediated diseases. Its unique mechanism of action, targeting complement component 3 (C3), makes it a versatile therapeutic candidate for a wide range of conditions beyond PNH. Ongoing clinical trials are exploring its efficacy in geographic atrophy, C3 glomerulopathy, and autoimmune hemolytic anemia, positioning Pegcetacoplan as a promising treatment for diseases driven by complement dysregulation.
In the case of geographic atrophy, a late-stage form of age-related macular degeneration, Pegcetacoplan is being tested for its ability to slow disease progression by inhibiting local complement activation in the retina. Early phase II results from the FILLY study demonstrated that intravitreal Pegcetacoplan significantly reduced the growth of geographic atrophy lesions. This trial’s positive outcomes have led to ongoing phase III studies, GALE, DERBY, and OAKS, which are investigating the long-term effects of Pegcetacoplan in this patient population.
Pegcetacoplan therapy for patients with PNH has been shown to have several benefits compared to treatment with other US FDA-approved therapies. In contrast to the requirement for intravenous dosing of eculizumab and ravulizumab, pegcetacoplan can be self-administered at home by the patient or a caregiver at their convenience (under guidance/training from a healthcare professional); thus eliminating the continuous need for an appointed nurse trained in intravenous drug administration and regular infusion clinic visits. In addition to improved hematologic parameters, patients receiving pegcetacoplan have reported increased QoL measures (such as decreased fatigue levels) in various clinical trials of the drug. A recent cost analysis demonstrated $1.7 million in cost savings for one US health insurance payer over a 3-year period due to reduced transfusion requirements and fewer breakthrough hemolysis events when patients were treated with pegcetacoplan.
These ongoing trials underscore Pegcetacoplan’s versatility and potential to become a cornerstone treatment across multiple complement-mediated diseases. As research continues, its role in expanding therapeutic options for patients will likely grow.
Pegcetacoplan: Transforming Complement Inhibition and Clinical Outcomes
Pegcetacoplan represents a transformative advancement in the treatment of complement-mediated diseases, offering a novel approach by inhibiting complement component 3 (C3). By targeting C3, Pegcetacoplan prevents both intravascular and extravascular hemolysis, addressing limitations seen with traditional C5 inhibitors. Its approval for paroxysmal nocturnal hemoglobinuria (PNH) has shown significant clinical benefits, including improved hemoglobin levels and reduced transfusion needs, positioning it as a superior therapy for patients with complement dysregulation.
Beyond PNH, ongoing research into Pegcetacoplan’s applications in other diseases, such as geographic atrophy, C3 glomerulopathy, and autoimmune hemolytic anemia, highlights its versatility. Clinical trials have demonstrated promising results in reducing disease progression and improving patient outcomes in these conditions. As research continues to evolve, Pegcetacoplan is likely to become a cornerstone in managing a wide range of complement-related disorders.
Overall, Pegcetacoplan’s broad therapeutic potential and its success in clinical trials mark it as a pivotal development in immunology, offering new hope for patients affected by complement-mediated diseases and significantly expanding treatment options in this field.
 LinkPeptide
LinkPeptide