Trofinetide: A Breakthrough in the Treatment of Rett Syndrome and Neurodevelopmental Disorders
Abstract
Trofinetide, a synthetic analog of glycine-proline-glutamate (GPE), represents a significant advancement in the treatment of neurodevelopmental disorders, particularly Rett syndrome. Developed by Neuren Pharmaceuticals and Acadia Pharmaceuticals, Trofinetide was approved by the FDA in March 2023, marking a milestone in addressing the unmet therapeutic needs of Rett syndrome patients. Its mechanism of action involves improving synaptic density and stabilizing cortical plasticity, though its precise effects remain under investigation. Clinical trials, such as the LAVENDER and DAFFODIL studies, have demonstrated the efficacy of Trofinetide in improving core symptoms of Rett syndrome. Despite common adverse events like diarrhea and weight loss, the treatment’s potential benefits are notable. As the first approved treatment for Rett syndrome, Trofinetide opens new avenues for research in neurodevelopmental therapies and holds promise for future applications in related disorders, such as Fragile X syndrome[1].
Introduction to Trofinetide: A Breakthrough in Neurodevelopmental Treatment
Trofinetide, a novel synthetic analog of glycine-proline-glutamate (GPE), has emerged as a breakthrough in treating neurodevelopmental disorders, particularly Rett syndrome. Developed by Neuren Pharmaceuticals and Acadia Pharmaceuticals, Trofinetide gained approval from the U.S. FDA in March 2023 as the first treatment specifically for Rett syndrome, addressing a significant unmet need in this population[1]. Rett syndrome is a rare and severe neurodevelopmental disorder caused primarily by mutations in the MECP2 gene, leading to impaired neurological development, severe cognitive deficits, and loss of motor functions[2]. Historically, treatments have focused on symptom management rather than targeting the underlying disease mechanisms, leaving many patients with limited options for effective interventions[3].
As the first approved treatment for Rett syndrome, Trofinetide opens new avenues for research in neurodevelopmental therapies and holds promise for future applications in related disorders, such as Fragile X syndrome. Its ability to modulate synaptic density and improve neurological function positions it as a key candidate for expanding research into other neurodevelopmental conditions where synaptic dysfunction plays a central role. Trofinetide’s approval marks a significant milestone in the ongoing search for effective treatments for rare pediatric neurological disorders.
Understanding Trofinetide’s Mechanism: How It Targets Neurological Deficits
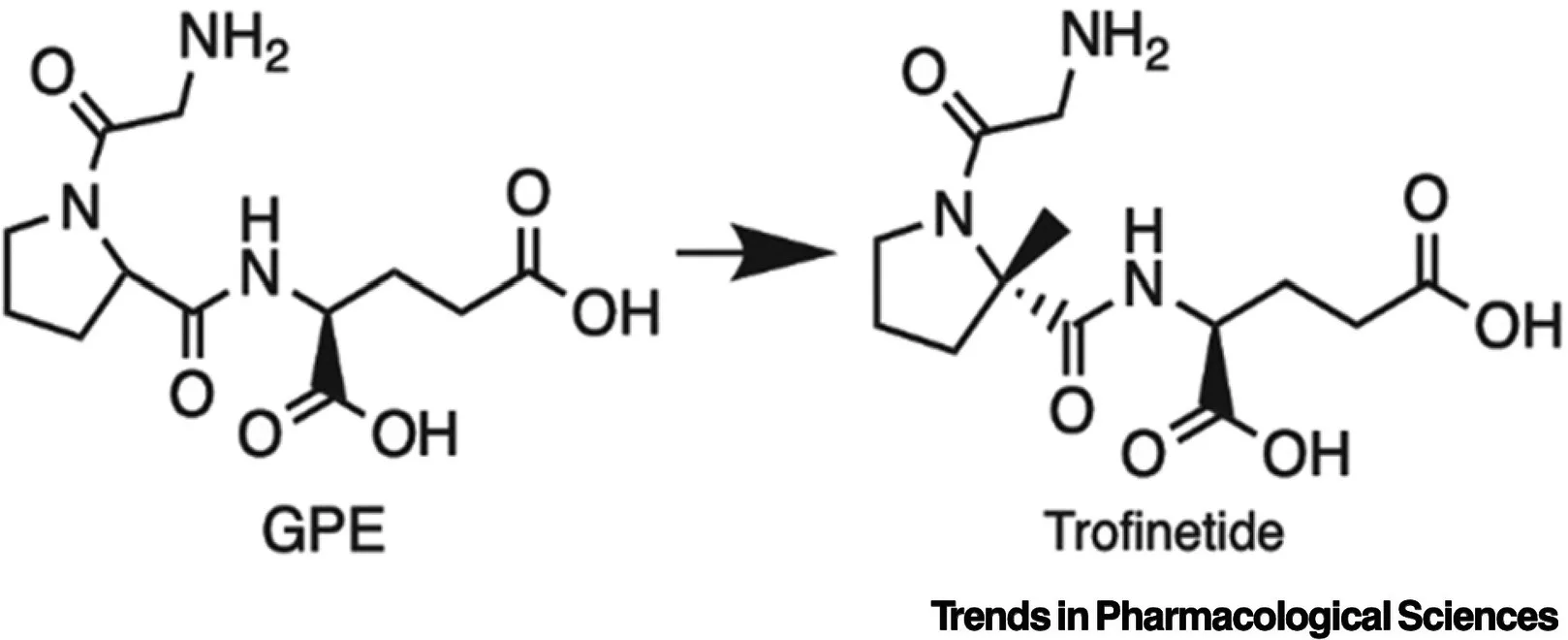
Trofinetide’s pharmacodynamics is centered on its role as a synthetic analog of glycine-proline-glutamate (GPE), a naturally occurring N-terminal tripeptide of insulin-like growth factor 1 (IGF-1). GPE is known for its neurotrophic and neuroprotective properties, which make it crucial in the development and maintenance of normal brain function[4]. Trofinetide has been engineered to have improved bioavailability and a longer elimination half-life compared to GPE, enhancing its therapeutic potential[5]. Its pharmacological effects are primarily observed in neurodevelopmental disorders, particularly Rett syndrome.
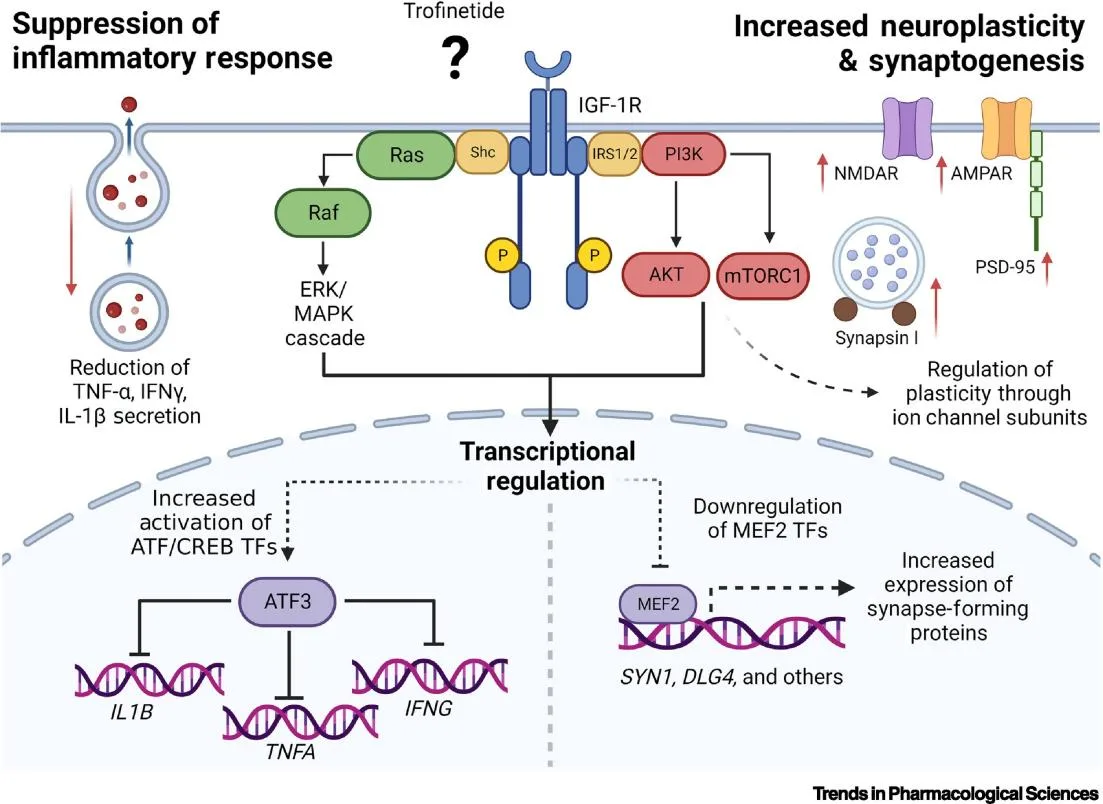
Although the exact mechanism of action in Rett syndrome remains unclear, preclinical studies have shown that Trofinetide has the potential to restore synaptic density and increase dendritic branching in animal models[6]. These effects suggest that Trofinetide can help stabilize neuronal function and plasticity, which are compromised in disorders like Rett syndrome. The drug’s ability to cross the blood-brain barrier and its neurotrophic properties further contribute to its therapeutic promise. As ongoing studies continue to explore its efficacy in both Rett syndrome and other neurodevelopmental conditions, Trofinetide stands as a significant advancement in neurotherapeutic treatments.
Clinical Efficacy of Trofinetide: Insights from Key Trials
Trofinetide’s efficacy in treating Rett syndrome has been rigorously evaluated in several clinical trials, with significant findings from Phase II and III studies. The LAVENDER trial, a Phase III randomized, double-blind, placebo-controlled study, was pivotal in demonstrating Trofinetide’s ability to improve core symptoms of Rett syndrome in girls and women aged 5-20 years. In this trial, patients who received Trofinetide showed significant improvements in the Rett Syndrome Behavior Questionnaire (RSBQ) total score and the Clinical Global Impression-Improvement (CGI-I) score compared to the placebo group. Additionally, the DAFFODIL trial, which focused on children aged 2-4 years, provided further evidence of Trofinetide’s efficacy in younger patients, reinforcing its therapeutic potential across age groups[7].
The therapeutic benefits observed in these trials include improvements in communication, social engagement, and overall quality of life. Notably, the LILAC open-label extension study demonstrated sustained improvements in these core symptoms over an extended treatment period, highlighting the drug’s long-term benefits[8]. These trials have established Trofinetide as a promising treatment for Rett syndrome, making it the first drug to directly target the underlying neurological deficits associated with this disorder.
Evaluating the Safety Profile of Trofinetide: Managing Adverse Events
While Trofinetide has shown therapeutic efficacy in treating Rett syndrome, it is associated with some adverse events that need careful management. The most commonly reported side effect in clinical trials was diarrhea, which affected up to 85% of patients receiving Trofinetide in the LAVENDER trial. Although diarrhea was generally mild to moderate in severity, some patients required dose adjustments or antidiarrheal treatments to manage symptoms. Other frequent adverse events included vomiting, decreased appetite, and fatigue, with vomiting occurring in about 29% of Trofinetide recipients compared to 12% in the placebo group.
Weight loss was another notable concern, with a significant percentage of patients experiencing a reduction in body weight during treatment. Around 12% of patients treated with Trofinetide had weight loss exceeding 7% of their baseline weight, prompting some to discontinue treatment. Despite these adverse events, the overall safety profile of Trofinetide is considered acceptable, particularly given the significant unmet need for effective treatments in Rett syndrome. Ongoing clinical monitoring and careful dose management help mitigate these risks, ensuring that patients can benefit from the drug while minimizing potential side effects.
Trofinetide’s FDA Approval and Future Potential in Neurotherapeutics
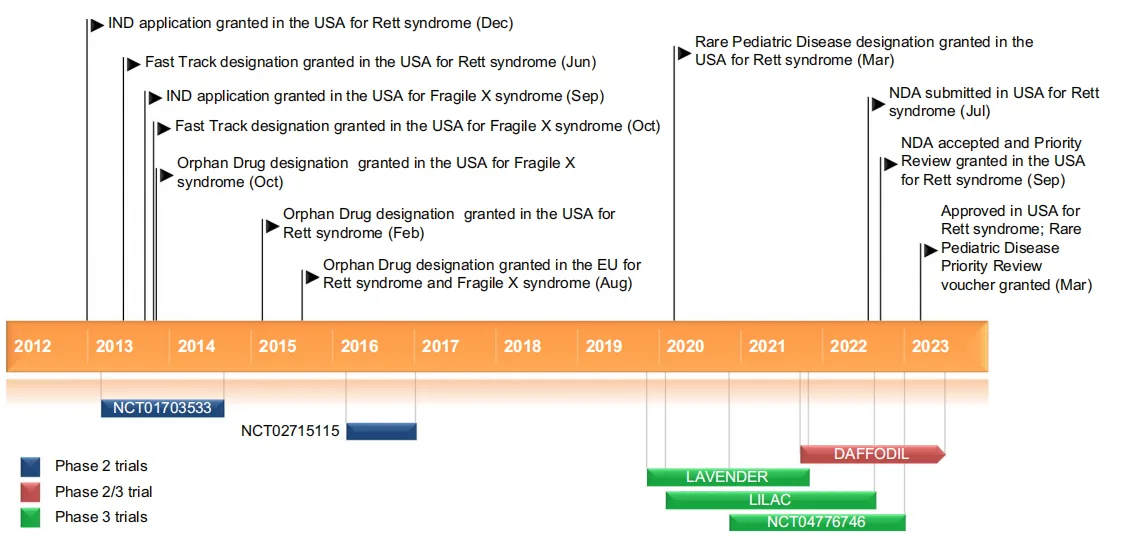
Trofinetide received its first regulatory approval from the U.S. Food and Drug Administration (FDA) in March 2023 for the treatment of Rett syndrome in adult and pediatric patients aged two years and older, marking a significant milestone in neurodevelopmental disorder therapies. This approval came after the successful completion of clinical trials, including the pivotal Phase III LAVENDER trial, which demonstrated the drug’s efficacy in improving core symptoms of Rett syndrome. The drug was granted Priority Review and Orphan Drug designation by the FDA due to the rarity of the condition and the significant unmet medical need.
Looking ahead, Trofinetide shows promise for expanded applications in other neurodevelopmental disorders, such as Fragile X syndrome. Ongoing clinical trials are investigating the efficacy of Trofinetide in younger patients and exploring its potential for treating a broader spectrum of cognitive and behavioral symptoms associated with neurodevelopmental conditions. The drug’s success may inspire future research into similar treatments that address the underlying biological mechanisms of rare neurological diseases. Additionally, the global market potential of Trofinetide could grow as it gains approvals in other regions and continues to be explored for other therapeutic indications.
Reference
- Collins, B. E., & Neul, J. L. (2022). Rett syndrome and MECP2 duplication syndrome: disorders of MeCP2 dosage. Neuropsychiatric disease and treatment, 2813-2835.
- Fu, C., Armstrong, D., Marsh, E., Lieberman, D., Motil, K., Witt, R., … & Benke, T. (2020). Consensus guidelines on managing Rett syndrome across the lifespan. BMJ paediatrics open, 4(1).
- Kyle, S. M., Vashi, N., & Justice, M. J. (2018). Rett syndrome: a neurological disorder with metabolic components. Open biology, 8(2), 170216.
- Silva-Reis, S. C., Sampaio-Dias, I. E., Costa, V. M., Correia, X. C., Costa-Almeida, H. F., García-Mera, X., & Rodríguez-Borges, J. E. (2023). Concise overview of glypromate neuropeptide research: from chemistry to pharmacological applications in neurosciences. ACS Chemical Neuroscience, 14(4), 554-572.
- Tropea, D., Giacometti, E., Wilson, N. R., Beard, C., McCurry, C., Fu, D. D., … & Sur, M. (2009). Partial reversal of Rett Syndrome-like symptoms in MeCP2 mutant mice. Proceedings of the National Academy of Sciences, 106(6), 2029-2034.
- Deacon, R. M., Glass, L., Snape, M., Hurley, M. J., Altimiras, F. J., Biekofsky, R. R., & Cogram, P. (2015). NNZ-2566, a novel analog of (1–3) IGF-1, as a potential therapeutic agent for fragile X syndrome. Neuromolecular medicine, 17, 71-82.
- Percy, A., Ryther, R., Marsh, E., Feyma, T., Lieberman, D., Neul, J., … & Youakim, J. (2023). Trofinetide for the treatment of Rett syndrome: an open-label study in girls 2 to 4 years of age (P13-9.005). Neurology, 100(17_supplement_2), 1378.
- Glaze, D. G., Neul, J. L., Kaufmann, W. E., Berry-Kravis, E., Condon, S., Stoms, G., … & Rett 002 Study Group. (2019). Double-blind, randomized, placebo-controlled study of trofinetide in pediatric Rett syndrome. Neurology, 92(16), e1912-e1925.
 LinkPeptide
LinkPeptide