Peptide Amphiphiles and Gene Therapy: Why Amorphous Nanostructures Outperform Fibrils
A New Twist in Viral Gene Delivery
Gene therapy holds the promise of treating diseases at their source by rewriting faulty genetic instructions. Central to this approach are retroviral vectors—viruses repurposed to deliver therapeutic genes into human cells. Despite their potential, one major hurdle remains: getting these vectors to efficiently attach and enter target cells. For years, researchers have relied on peptide-based enhancers built around rigid fibrillar structures to improve this process. But a new study reveals a surprising alternative. Instead of order and rigidity, amorphous peptide nanostructures may offer the key to safer, more effective viral gene delivery.
Why Today’s Viral Boosters Fall Short
To boost viral entry, scientists have designed transduction enhancers—materials that help viruses stick to cells more effectively. Among the most studied are self-assembling peptides that form long, fibrillar nanostructures. These peptide fibrils work by reducing electrostatic repulsion between the negatively charged virus and the equally negative cell membrane, essentially acting as a molecular bridge. While effective, this approach has limitations. Viruses often become trapped inside dense fibril networks instead of reaching the cell surface, reducing efficiency. Even more concerning, fibrillar assemblies can be slow to degrade, raising questions about safety and potential toxicity. These drawbacks highlight the urgent need for alternatives that combine strong viral attachment with biocompatibility.
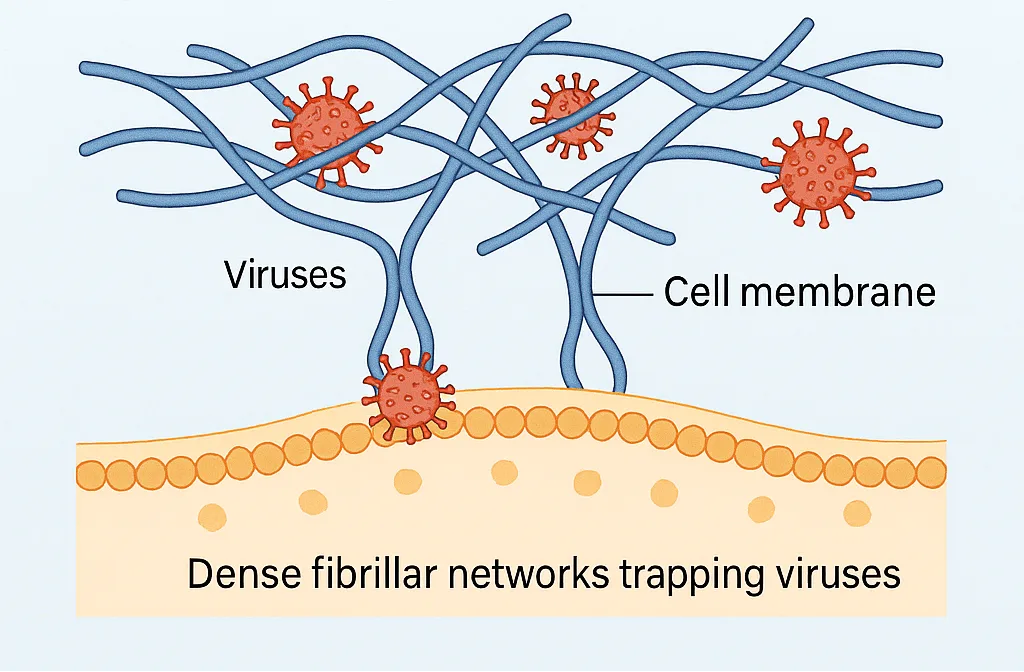
Peptide Amphiphiles: Small Molecules, Big Potential
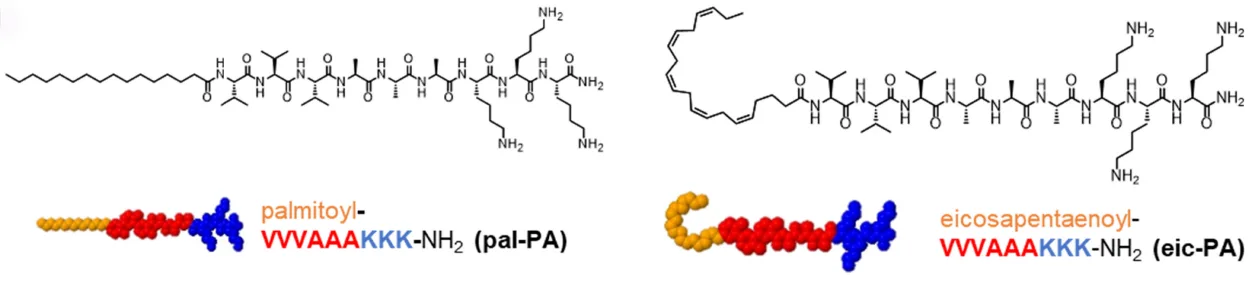
Peptide amphiphiles (PAs) are a fascinating class of hybrid molecules made by linking a peptide sequence to a lipid tail. This simple design gives them the ability to self-assemble into nanostructures with unique biological properties. In the new study, researchers compared two versions: pal-PA, built with a saturated palmitoyl lipid, and eic-PA, which carries an unsaturated fatty acid known as eicosapentaenoic acid. The difference in lipid chemistry leads to very different behaviors. Pal-PA forms the expected rigid fibrils, while eic-PA assembles into irregular, amorphous aggregates. The unsaturated chain in eic-PA prevents tight molecular packing, introducing flexibility and disorder. This structural shift proved critical, as it reshaped how viruses and cells interact with these materials.
Microscopy Reveals a Surprising Advantage
Using advanced imaging techniques, the team compared how pal-PA and eic-PA behave at the nanoscale and during virus–cell interactions. As expected, pal-PA formed long, fibrillar networks, while eic-PA assembled into countless small, spherical, amorphous clusters. In fact, eic-PA produced nearly five times more aggregates than pal-PA, and these clusters were significantly smaller in size.
When tested with virus-like particles (VLPs) and human cells, the differences became striking. Pal-PA’s fibrils tended to trap VLPs inside their mesh-like structures, limiting the particles’ ability to make direct contact with the cell membrane. By contrast, eic-PA’s amorphous fragments coated VLPs and helped them attach directly to the plasma membrane and cellular protrusions, such as filopodia. Microscopy images even revealed smoother cell surfaces with pal-PA, compared to eic-PA, where tiny fragments seemed to cover both the virus and the cell.
This ability to create a “molecular bridge” between virus and cell gives eic-PA a distinct advantage, making viral entry more efficient and paving the way for superior gene transfer.
When Disorder Outperforms Order
The study highlights a fundamental shift in design philosophy: disorder can be more effective than order. Fibrillar peptide assemblies, like those formed by pal-PA, are rigid and structured, but this rigidity limits accessibility for viruses and slows biodegradation. In contrast, amorphous assemblies created by eic-PA are smaller, more numerous, and more flexible, offering a larger surface area for interaction. This means viruses are not trapped within a network but are instead guided straight to the cell membrane. Just as important, eic-PA showed nearly complete biodegradability (99%), a key factor for clinical safety. Together, these advantages make amorphous nanostructures a promising new class of enhancers for viral gene delivery.
Transforming the Future of Gene Therapy
For gene therapy, where safe and efficient delivery is everything, these findings are especially exciting. Amorphous peptide amphiphiles like eic-PA could serve as next-generation transduction enhancers, improving the way therapeutic genes are introduced into patient cells. By combining high efficiency with rapid biodegradability, they address the two biggest concerns in viral delivery: effectiveness and safety. Beyond retroviral vectors, this design principle may also apply to other delivery platforms, expanding the toolkit for researchers developing treatments for cancer, inherited disorders, and metabolic diseases. In short, amorphous nanostructures could help gene therapy reach its full clinical potential.
Redefining the Rules of Viral Enhancement
This study challenges the long-standing belief that rigid fibrils are the best way to boost viral entry. Instead, it shows that flexible, amorphous nanostructures can outperform them—delivering viruses more efficiently while breaking down safely in the body. As research continues, peptide amphiphiles like eic-PA may redefine how we design biomaterials for the future of gene therapy.
Reference
La Roche, J., Rauch‐Wirth, L., Zimmerman, L., Zech, F., Münch, J., Read, C., & Kaygisiz, K. (2025). Interactions of Peptide Amphiphiles With Viruses and Cells Are Enabled by Amorphous Nanostructures. Journal of Peptide Science, 31(9), e70051.
https://doi.org/10.1002/psc.70051
Morgan, M. A., Galla, M., Grez, M., Fehse, B., & Schambach, A. (2021). Retroviral gene therapy in Germany with a view on previous experience and future perspectives. Gene Therapy, 28(9), 494-512.
https://doi.org/10.1038/s41434-021-00237-x
Sieste, S., Mack, T., Lump, E., Hayn, M., Schütz, D., Röcker, A., … & Weil, T. (2021). Supramolecular peptide nanofibrils with optimized sequences and molecular structures for efficient retroviral transduction. Advanced Functional Materials, 31(17), 2009382.
https://doi.org/10.1002/adfm.202009382
Yang, S., & Dong, H. (2020). Modular design and self-assembly of multidomain peptides towards cytocompatible supramolecular cell penetrating nanofibers. RSC advances, 10(49), 29469-29474.
 LinkPeptide
LinkPeptide