Precision Oncology Reinvented: How Anticancer Peptides Are Changing the Game
Abstract
Anticancer peptides (ACPs) are emerging as a promising frontier in oncology, offering a novel approach to cancer treatment that is both targeted and versatile. Unlike traditional therapies that often damage healthy tissue, ACPs selectively attack cancer cells through mechanisms such as membrane disruption, apoptosis induction, and immune modulation. Derived from natural sources or engineered synthetically, these peptides are being refined through chemical modifications, non-natural amino acids, and advanced delivery systems like nanoparticles and gene vectors. Moreover, artificial intelligence and molecular docking are accelerating peptide discovery and optimization, helping fast-track their journey from computer modeling to clinical trials. With several ACPs already under investigation in human studies, this new class of therapeutics holds significant promise for improving treatment precision, reducing side effects, and overcoming resistance. As research advances, ACPs may well become essential components of next-generation cancer therapies.
Why Cancer Needs Smarter Weapons: Enter Anticancer Peptides
Cancer remains one of the most persistent and deadly health challenges of the modern era, responsible for millions of deaths each year. While advances in chemotherapy, radiation, and immunotherapy have improved outcomes for many, these conventional treatments often come with significant drawbacks—ranging from severe side effects to the eventual development of drug resistance. In many cases, these therapies act like blunt-force tools, destroying both malignant and healthy cells, and leaving patients with compromised immunity and diminished quality of life.
In the pursuit of more refined and effective cancer treatments, researchers are turning to a promising class of molecules: anticancer peptides (ACPs). These short chains of amino acids—some naturally occurring in organisms like bees, frogs, and marine microbes—are now being engineered to target cancer cells with precision. What makes ACPs especially compelling is their ability to selectively attack tumor cells, disrupt their structure or metabolism, and in some cases, activate the body’s immune defenses, all while minimizing damage to normal tissues.
In this blog, we’ll explore the rise of anticancer peptides: what they are, how they work, how scientists are optimizing them through cutting-edge technologies, and why they may soon play a pivotal role in the next generation of cancer therapies.
Breaking Down ACPs: What They Are and Why They Matter
Anticancer peptides (ACPs) are a specialized class of therapeutic molecules composed of short chains of amino acids, typically ranging from 10 to 100 residues. Unlike conventional chemotherapeutic agents, which often lack selectivity, ACPs are uniquely designed—or naturally evolved—to target cancer cells while sparing healthy ones. Their defining characteristics include a positive net charge and amphipathic structure, allowing them to interact efficiently with the negatively charged membranes of cancer cells.
Sources and Origins
ACPs can be derived from a wide array of natural sources:
- Animals: Peptides like melittin from bee venom and magainin from frogs exhibit strong anticancer activity.
- Microbes: Bacteriocins from bacteria are known for their selective cytotoxicity.
- Plants: Cyclotides from species like Viola arvensis show robust antitumor effects.
- Synthetic design: Advances in peptide chemistry have enabled the creation of novel ACPs with enhanced stability and potency.
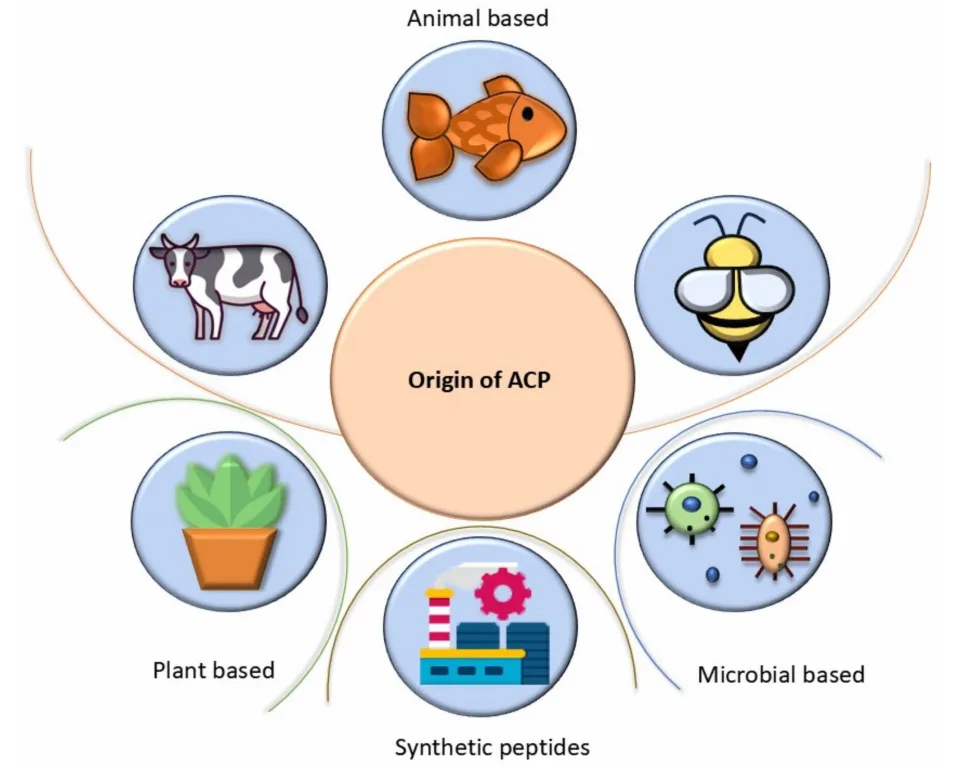
Each of these sources offers unique structural scaffolds, enabling scientists to modify and optimize their sequences for improved therapeutic outcomes.
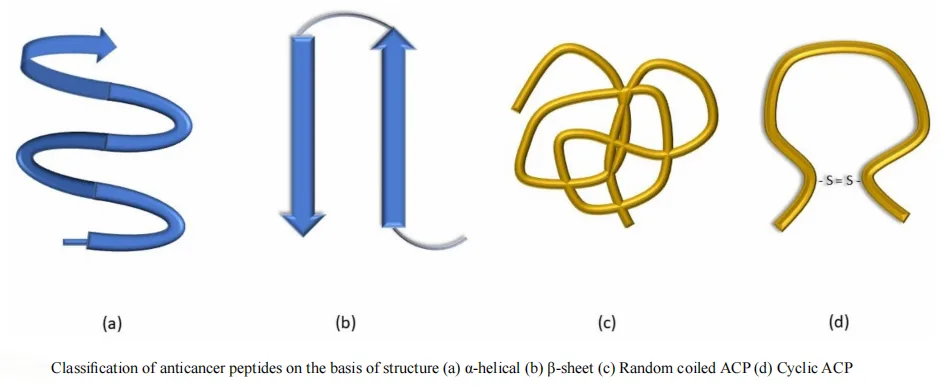
Structural Classification
ACPs are commonly classified based on their secondary structure:
- α-Helical Peptides: The most prevalent class, often derived from amphibians (e.g., Aurein 1.2, BMAP-28). Known for potent membrane-disruptive activity.
- β-Pleated Sheet Peptides: Stabilized by disulfide bonds and often found in mammals and plants (e.g., LfcinB, HNP-1).
- Random Coil Peptides: Lacking a fixed secondary structure but rich in proline and glycine; offer low toxicity to normal cells (e.g., Alloferon, Anginex).
- Cyclic Peptides: Characterized by enhanced stability due to cyclization or disulfide bonds; show strong resistance to enzymatic degradation and are often isolated from plants (e.g., RA-V, Diffusa Cytides).
Why ACPs Are Different
What sets ACPs apart is not just their origin or structure, but their high degree of specificity, lower risk of resistance, and versatility in function. They can disrupt cancer cell membranes, interfere with intracellular targets, modulate immune responses, and even serve as delivery vehicles for other drugs.
As we’ll explore next, their mechanisms of action are as varied as they are powerful—paving the way for more selective, personalized cancer treatments.
Precision Strikes: How Anticancer Peptides Eliminate Tumors
One of the most compelling features of anticancer peptides (ACPs) is their diverse and highly targeted modes of action. Unlike traditional therapies that broadly target rapidly dividing cells—often harming healthy tissues in the process—ACPs leverage the unique biophysical and biochemical properties of cancer cells to attack them with precision. Their mechanisms range from disrupting cell membranes to triggering programmed cell death and blocking tumor growth signals.
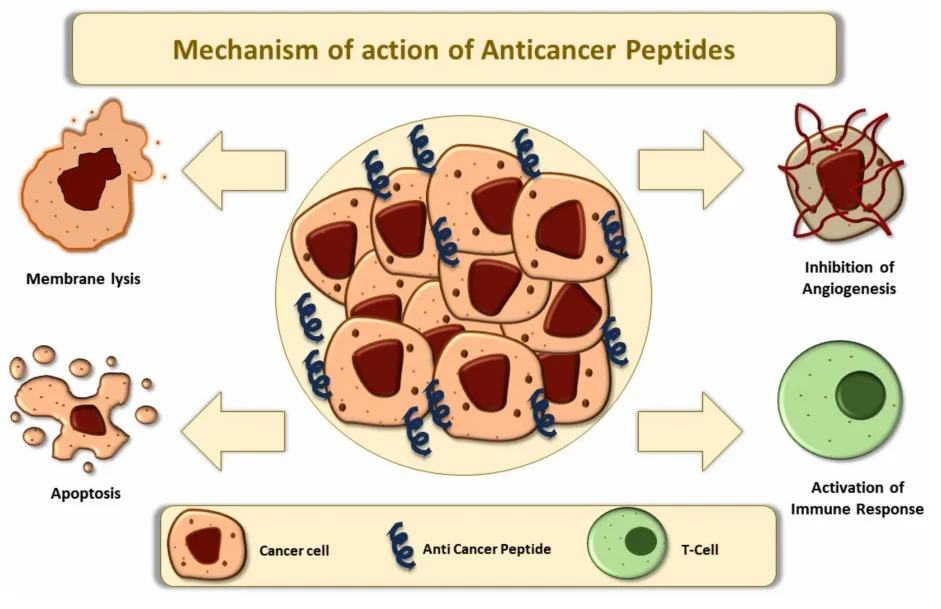
1. Membrane Disruption: The First Line of Attack
Many ACPs exploit a key vulnerability in cancer cells: their negatively charged cell membranes. This charge difference, largely absent in healthy cells, allows positively charged ACPs to bind preferentially to cancer cells. Once attached, ACPs can disrupt the membrane in several ways:
- Barrel-Stave Model: Peptides insert themselves into the membrane and form barrel-like pores that puncture the cell, leading to leakage and death.
- Toroidal Pore Model: ACPs bend the membrane into a continuous curve, creating unstable pores that compromise cell integrity.
- Carpet Model: At high concentrations, ACPs spread across the membrane like a detergent, causing wholesale rupture.
Examples include temporins, melittin, and LL-37, which have demonstrated potent cytolytic effects through these pathways.
2. Apoptosis: Programming Cell Death
Beyond membrane disruption, many ACPs initiate apoptosis, a form of programmed cell death that eliminates cancer cells without triggering inflammation.
- ACPs like D(KLAKLAK)₂ and Baceridin disrupt the mitochondrial membrane, leading to the release of cytochrome c and activation of caspases—enzymes that orchestrate cellular breakdown.
- Some ACPs, such as Melittin, modulate key signaling pathways like ERK and Akt, making them highly effective in inducing apoptosis in resistant tumor cells.
Because apoptosis is a clean, non-inflammatory process, ACPs that use this route offer a therapeutic advantage by avoiding collateral tissue damage.
3. Beyond Cell Death: Targeting Tumor Growth and Immunity
ACPs don’t stop at killing individual cancer cells—they can also interfere with the tumor environment:
- Anti-angiogenesis: Peptides like KV11 and Temporin-1CEa block the formation of new blood vessels, starving tumors of nutrients and oxygen.
- Immune modulation: ACPs such as Alloferon and LfcinB activate natural killer (NK) cells and enhance T-cell responses, boosting the body’s innate ability to fight cancer.
- Hormonal interference: Some peptides regulate cancer-promoting hormones, such as FSH and GHRH, helping suppress tumor growth at the systemic level.
In short, ACPs work on multiple fronts—physically damaging cancer cells, disrupting internal processes, and altering the tumor microenvironment—all with remarkable precision.
Rewriting Nature: How Scientists Are Enhancing Peptide Power
While many anticancer peptides (ACPs) are derived from nature, their true therapeutic potential is being unlocked through biochemical engineering and smart delivery technologies. Scientists are optimizing these molecules to enhance their stability, specificity, and safety, paving the way for clinical applications that overcome the limitations of traditional cancer treatments.
Chemical Modifications: Small Tweaks, Big Impact
ACPs can be chemically modified to improve their performance in the human body. These alterations are typically made to either the main peptide chain or the side chains:
- Main Chain Modifications: By substituting specific amino acids, researchers can fine-tune a peptide’s hydrophobicity, charge, and folding behavior. For example, analogs of the K7S peptide have been modified to reduce toxicity while maintaining anticancer activity.
- Side Chain Modifications:
- PEGylation: Attaching polyethylene glycol (PEG) to peptides increases their half-life and reduces immune detection.
- Glycosylation and Phosphorylation: These natural post-translational modifications can improve solubility and receptor binding.
- Cholesterol Conjugation: Enhances membrane interaction and stability.
These enhancements make ACPs more suitable for in vivo use, allowing for lower dosages and fewer side effects.
Synthetic Strategies and Non-Natural Amino Acids
To further boost specificity and resist enzymatic degradation, researchers are designing peptides using D-amino acids and non-natural residues. These synthetic building blocks are not recognized by the body’s enzymes, making the peptides more stable and bioavailable. One example is D-K6L9, a peptide that retains its anticancer function while minimizing toxicity to healthy tissues.
Incorporating these strategies ensures that ACPs maintain activity in the bloodstream and reach their tumor targets intact.
Advanced Delivery Systems
Even the most potent peptides require an effective delivery mechanism. Enter nanotechnology, which offers precise control over where and how a peptide is released:
- Liposomes and Nanoparticles: These carriers can encapsulate peptides, protect them from degradation, and deliver them directly to tumor sites.
- Polymeric Conjugates: Help control the rate of peptide release and improve pharmacokinetics.
- Gene Therapy Vectors: Deliver DNA or mRNA that encodes the ACP, allowing the body to produce the peptide at the tumor site itself.
These delivery innovations are transforming ACPs into clinically viable, targeted therapeutics, reducing systemic toxicity and improving treatment outcomes.
Designed by Algorithms, Tested in Humans: The Journey of ACPs
The development of anticancer peptides (ACPs) has been supercharged by artificial intelligence (AI) and computational modeling, allowing scientists to design, screen, and optimize peptides with unprecedented speed and precision.
AI-driven tools like AntiCP, ACPred, and pACP-HybDeep analyze peptide sequences to predict anticancer activity, structural stability, and toxicity. Advanced models such as ACP-ESM, which leverage transformer-based algorithms, can even forecast how peptides will behave in complex biological environments—before they’re ever synthesized in the lab.
Complementing this is molecular docking, which simulates how peptides interact with cancer-related proteins, helping researchers identify the most promising candidates for preclinical testing.
These technologies have already paved the way for several ACPs to enter clinical trials:
- LTX-315, a synthetic peptide that kills cancer cells and stimulates the immune system, has shown promise in melanoma.
- CIGB-300 and P28 target specific cancer-associated proteins and are under active investigation for multiple tumor types.
- Peptide-based vaccines like ImMucin are also gaining traction for their ability to trigger long-term immune responses against tumors.
With AI at the helm and clinical momentum building, ACPs are rapidly transitioning from experimental tools to real-world therapeutics poised to redefine cancer treatment.
 LinkPeptide
LinkPeptide