Reprogramming Immunity: How Khavinson Peptides Calm Inflammation and Support Regeneration
Abstract
Monocytes and macrophages are central players in the innate immune system, responsible for detecting threats, coordinating inflammation, and supporting tissue repair. But when these processes become overactive, they can fuel chronic inflammation and disease.
Recent research is shedding light on a novel class of regulatory molecules: Khavinson peptides. Originally isolated from animal tissues, these short peptides exhibit remarkable biological effects—including the ability to promote cell renewal and modulate immune responses. Unlike conventional immunomodulators, they act directly at the cellular level, bypassing receptor-dependent pathways.
In this blog, we explore groundbreaking findings on how five Khavinson peptides—Epitalon, Vilon, Thymogen, Thymalin, and Chonluten—interact with THP-1 cells, a human monocytic model. From enhancing proliferation to rebalancing cytokine production, these peptides offer a promising strategy for controlling immune activity without triggering excess inflammation. Their potential could redefine how we approach immune regulation in aging and chronic disease.
Fueling Immune Renewal: How Peptides Promote Safe and Balanced Cell Proliferation
Khavinson peptides are best known for their anti-aging and regenerative effects—but their role in cell proliferation, particularly within the immune system, is now coming into sharper focus. In the THP-1 monocytic cell line, all five studied peptides—Epitalon, Vilon, Thymogen, Thymalin, and Chonluten—stimulated cell growth without disrupting normal cell cycle progression.
The study found that these peptides enhance the activity of ERK1/2, a mitogen-activated protein kinase (MAPK) crucial for promoting DNA synthesis and cell division. Except for Vilon, all peptides significantly increased ERK1/2 phosphorylation, suggesting they activate intracellular pathways associated with proliferation. Additionally, p70 S6 kinase, a downstream target of mTOR signaling involved in protein synthesis and cell growth, was also activated—especially by Thymalin and Thymogen.
Interestingly, Chonluten showed a dual behavior: while it promoted proliferation, it also triggered a modest increase in apoptosis. This nuanced effect may reflect its origin from bronchial epithelial tissue, where immune cell turnover must be tightly regulated during inflammation.
The researchers also observed an increase in extracellular vesicle (EV) release, particularly with Thymalin. EVs carry molecular cargo such as proteins and RNA and serve as markers of active cell communication. Their elevation suggests that these peptides may not only boost immune cell numbers but also enhance their signaling capacity.
Together, these findings position Khavinson peptides as pro-regenerative agents capable of stimulating controlled immune cell expansion—an effect with potential applications in immunodeficiency, wound healing, and cellular rejuvenation therapies.
Taming the Fire: Precision Inflammation Control with Bioactive Peptides
While boosting immune cell proliferation is valuable, uncontrolled inflammation can be equally dangerous. A hallmark of many chronic and autoimmune conditions is the overproduction of pro-inflammatory cytokines—molecules like TNF-α, IL-6, and IL-17 that amplify immune responses and damage healthy tissue.
In this study, THP-1-derived macrophages were exposed to lipopolysaccharide (LPS), a bacterial toxin known to trigger robust inflammatory responses. The result was a surge in cytokine secretion—mimicking the kind of immune activation seen in sepsis or chronic inflammation. However, when the cells were co-treated with Khavinson peptides, a striking pattern emerged: inflammation was dampened across the board.
All five peptides significantly reduced the secretion of TNF-α and IL-6, two cytokines heavily implicated in systemic inflammation and tissue damage. Even IL-17, often associated with autoimmune pathology, was decreased—most notably by Thymogen, which showed the strongest suppressive effect on this cytokine.
Chonluten, while slightly increasing baseline TNF-α levels when used alone, appeared to moderate inflammatory signaling under LPS stimulation. This mild induction may reflect a tolerogenic effect, promoting immune readiness without triggering a full inflammatory cascade—a phenomenon sometimes referred to as TNF tolerance.
These results suggest that Khavinson peptides possess selective anti-inflammatory capabilities: they do not shut down the immune system but rather fine-tune its responses. By interfering with key signaling nodes and suppressing overactive cytokine release, they offer a way to control immune activity without the side effects of immunosuppressive drugs.
In the context of autoimmune diseases, chronic infections, or even age-related inflammation (“inflammaging”), these peptides may serve as a gentler, biologically compatible intervention—capable of rebalancing immune responses without compromising defense.
Signaling Without Side Effects: Rewiring Immune Responses via STAT Pathways
One of the most compelling discoveries from the study is how Khavinson peptides influence the STAT signaling pathways, which are central to immune cell behavior. Specifically, the focus was on STAT1 and STAT3, two transcription factors that respond to cytokines and growth signals.
Under normal conditions, STAT1 is activated through receptor-mediated pathways—often via interferon-α (IFN-α) binding to its cell surface receptor. However, in peptide-treated macrophages, STAT1 phosphorylation occurred independently of IFN-α signaling. Western blotting confirmed no increase in IFN-α production, yet STAT1 was strongly activated by Epitalon, Vilon, and Chonluten.
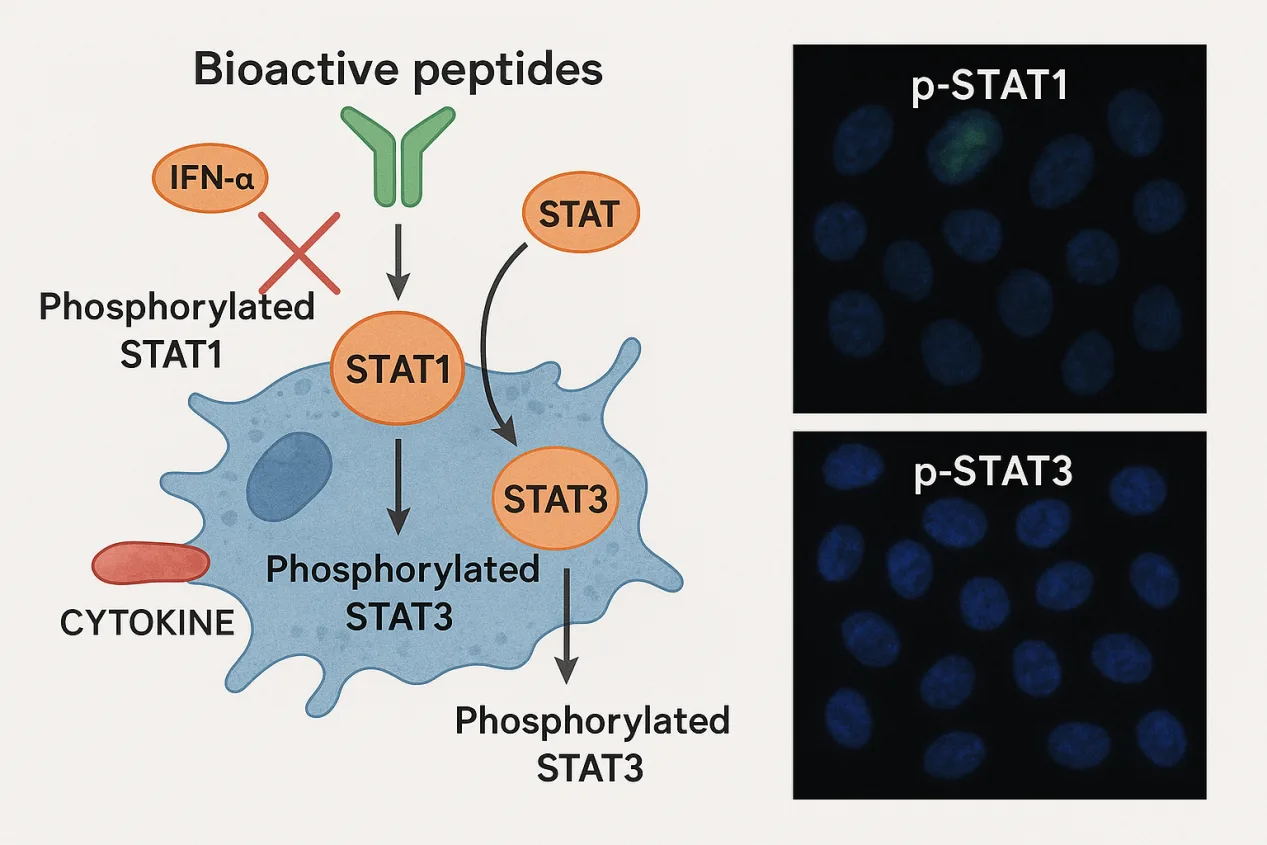
Confocal microscopy provided visual confirmation: phosphorylated STAT1 translocated into the nucleus, where it can initiate gene transcription linked to immune modulation and anti-inflammatory responses. This receptor-independent mechanism implies that these peptides can bypass typical extracellular signaling routes and act intracellularly, possibly by directly influencing transcriptional machinery.
In contrast, STAT3, which is heavily involved in acute inflammatory responses and IL-6 production, showed an opposite trend. Peptide treatment either had no effect or slightly downregulated STAT3 phosphorylation, even in the presence of LPS. This finding aligns with earlier results showing decreased IL-6 secretion and supports the idea that Khavinson peptides selectively silence pro-inflammatory pathways.
This dual action—activating STAT1 while suppressing STAT3—positions Khavinson peptides as precision modulators of immune signaling. Rather than broadly inhibiting immune function, they seem to reroute it toward a more balanced, anti-inflammatory profile, offering therapeutic promise for diseases driven by cytokine dysregulation.
Breaking the Chain: How Peptides Reduce Immune Cell Overactivation and Vascular Infiltration
Beyond controlling cytokine production, Khavinson peptides also influence how immune cells interact with the vascular system—a key step in inflammation. Normally, when blood vessels are activated by pro-inflammatory stimuli like LPS, they express adhesion molecules such as ICAM-1, which attract monocytes to exit circulation and infiltrate tissues.
To model this, the researchers used HUVECs (human umbilical vein endothelial cells) stimulated with LPS to mimic an inflamed vascular environment. Monocytes pretreated with Khavinson peptides were then added to these endothelial layers. The results were clear: cell adhesion was significantly reduced by all peptides, except for Thymalin.
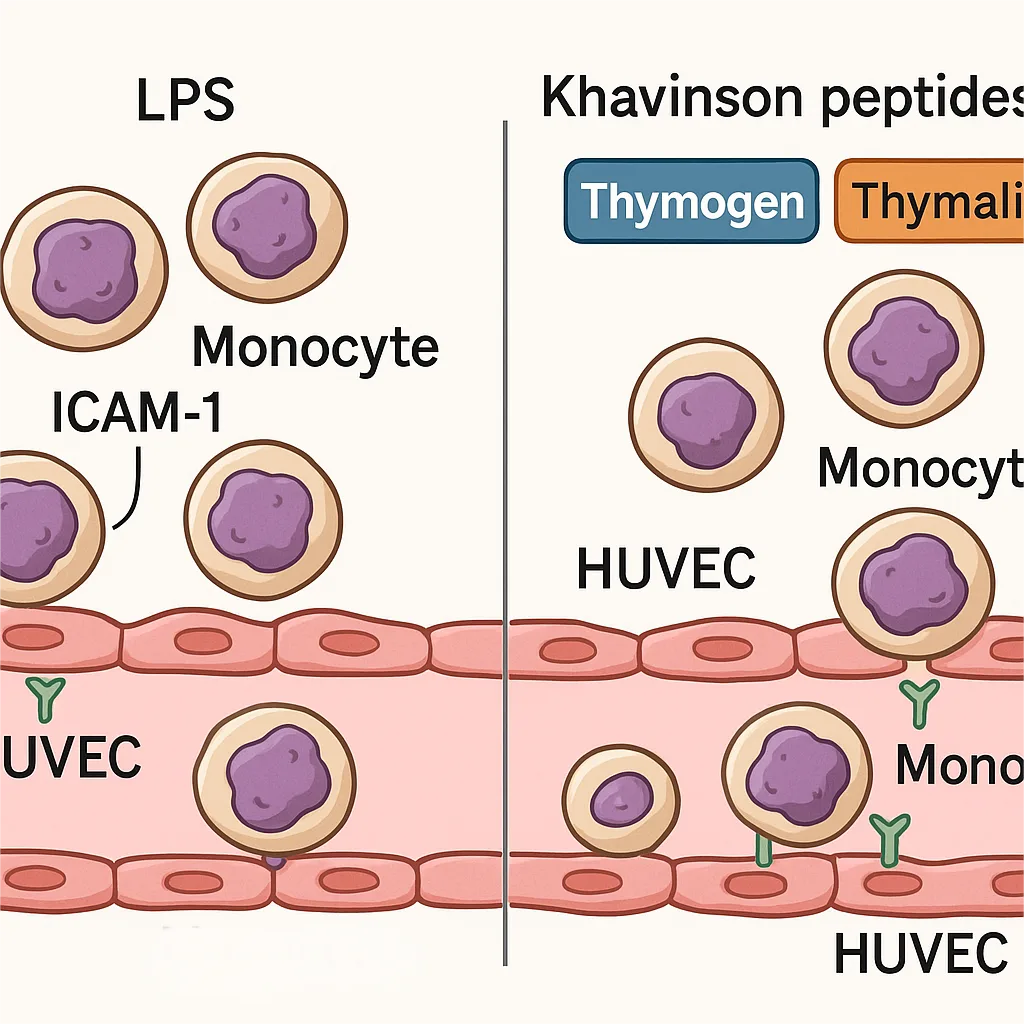
Thymogen showed the strongest inhibitory effect, bringing monocyte adhesion levels close to those seen in non-inflamed controls. This suggests a direct role for peptides in limiting immune cell recruitment, potentially preventing excessive tissue infiltration—a common problem in chronic inflammatory diseases.
Interestingly, Thymalin, a natural thymic extract, actually increased adhesion in this setup, possibly due to its complex composition containing a wider range of immune-modulating factors.
Altogether, this part of the study underscores a key takeaway: Khavinson peptides not only regulate internal cell signaling but also modulate how immune cells physically interact with their environment, adding another layer to their anti-inflammatory potential.
A New Class of Immune Modulators: Where Regeneration Meets Regulation
The findings from this study reveal a remarkable capacity of Khavinson peptides to act as multifaceted regulators of immune cell behavior. Through enhancing proliferation, selectively downregulating inflammatory cytokines, reprogramming STAT signaling, and reducing cell adhesion, these short-chain peptides orchestrate a balanced immune response without compromising cellular viability or function.
Their ability to act independently of traditional receptor pathways, especially in the modulation of STAT1 and cytokine output, highlights their therapeutic promise for conditions marked by immune imbalance—ranging from chronic inflammation and autoimmunity to age-related immune decline.
As biologically compatible molecules with a high safety profile, Khavinson peptides offer an innovative path toward immune modulation without immune suppression. While clinical translation will require further study, their performance in controlled in vitro settings like the THP-1 model makes a strong case for their potential.
In the landscape of future immunotherapies, these peptides may well become precision tools for restoring balance where it matters most.
 LinkPeptide
LinkPeptide