This Tiny Peptide Could Change How We Treat Alzheimer’s and Parkinson’s
Abstract
Humanin, a 24-amino acid mitochondrial-derived peptide, has emerged as a powerful neuroprotective agent with potential therapeutic applications across a spectrum of neurodegenerative disorders, including Alzheimer’s disease, Parkinson’s disease, and stroke. Discovered in 2001, Humanin protects neurons through multiple mechanisms—blocking apoptosis, enhancing mitochondrial function, and inhibiting amyloid-beta aggregation. Researchers have since developed potent analogues such as HNG, Colivelin, and HNSS, which exhibit enhanced stability, receptor binding, and therapeutic efficacy. These analogues have demonstrated success in preclinical models by improving cognition, reducing inflammation, and restoring mitochondrial health. Humanin also interacts with cell-surface and intracellular receptors, triggering survival pathways like STAT3 and ERK1/2. Despite challenges in delivery and mixed findings regarding its role in cancer, Humanin represents a promising class of bioactive peptides. Ongoing advances in peptide engineering and drug delivery systems could pave the way for Humanin-based therapeutics to enter clinical use in the battle against age-related brain diseases.
Unlocking the Power of Mitochondria: Humanin and the Future of Brain Health
Once thought of solely as the cell’s energy generators, mitochondria are now recognized as dynamic regulators of cell survival, signaling, and stress response. Among the most intriguing discoveries to emerge from this expanded view is a class of molecules known as mitochondrial-derived peptides (MDPs)—tiny, bioactive proteins with powerful protective properties. Leading this group is Humanin, a 24-amino acid peptide first identified in 2001 from the brain of an Alzheimer’s patient.

Humanin gained immediate scientific attention for its remarkable ability to shield neurons from damage caused by amyloid-beta (Aβ) and other Alzheimer’s-related toxins. Since then, its influence has proven to extend far beyond Alzheimer’s disease. Studies now show that Humanin and its synthetic analogues may offer therapeutic potential for a range of neurodegenerative conditions, including Parkinson’s disease, stroke, and age-related cognitive decline.
What sets Humanin apart is not just its neuroprotective strength, but its versatility—it targets multiple cellular pathways involved in inflammation, apoptosis, and mitochondrial dysfunction. In this article, we’ll explore the discovery and function of Humanin, examine the advances made through its synthetic derivatives, and look ahead to how this family of mitochondrial peptides may reshape the future of brain health and neurodegenerative disease treatment.
Meet Humanin: A Mitochondrial Messenger with a Mission
Humanin was first discovered during a cDNA screening of brain tissue from an individual with Alzheimer’s disease. Surprisingly, this neuroprotective factor was found in the relatively unaffected occipital lobe, hinting at its role as a natural cellular defender. The name “Humanin” reflects its potential to restore “humanity” to patients suffering from neurodegenerative decline.
What truly distinguishes Humanin is its mitochondrial origin. It is encoded within the mitochondrial 16S rRNA gene (MTRNR2)—a rare example of a biologically active peptide produced by mitochondrial DNA. This makes it part of the growing class of mitochondrial-derived peptides (MDPs), which includes others like MOTS-c and SHLPs that play key roles in cellular health and longevity.
Structurally, Humanin features:
- A positively charged N-terminal (MAPR)
- A hydrophobic core (GFSCLLLLTSEIDL)
- A polar C-terminal tail (PVKRRA)
This combination gives Humanin the flexibility to adopt different conformations depending on its environment, including an α-helical structure in less polar conditions. Several key residues—including S7, C8, and L9–L12—are critical to its neuroprotective, anti-apoptotic, and dimerization functions.
Though just 24 amino acids long, Humanin represents a breakthrough in mitochondrial signaling and therapeutic peptide design.
Smarter, Stronger, Sharper: The Rise of Humanin Analogues
To amplify the protective effects of native Humanin, researchers have developed a range of synthetic analogues—modifying its structure to enhance potency, resist degradation, and improve cellular delivery.
The standout performer is HNG, where serine at position 14 is replaced with glycine (S14G). This single substitution increases neuroprotective activity by a factor of 1000, making it one of the most potent Humanin derivatives to date. HNG has shown remarkable efficacy in animal models of Alzheimer’s, improving cognition and reducing amyloid burden.

Another significant variant is HN-D-Ser14, which incorporates a D-form serine at the same position. This not only resists enzymatic breakdown but also increases binding affinity to amyloid-beta, helping prevent toxic plaque formation.
Other innovative analogues include:
- Colivelin (CL): a fusion of Humanin with an activity-dependent neurotrophic factor, offering potent protection even at femtomolar concentrations.
- tHN-C3: a modified peptide that crosses biological barriers and inhibits caspase-3, a key driver of apoptosis.
- HNSS: a hybrid combining HNG with SS31, a mitochondria-targeting antioxidant, offering synergistic benefits for brain and mitochondrial health.
These analogues demonstrate how smart peptide engineering can enhance therapeutic potential—each version fine-tuned for specific roles in neuroprotection and cell survival.
How Humanin Fights Back: Inside Its Neuroprotective Arsenal
Humanin’s strength lies in its multi-layered mode of action, operating both inside and outside the cell to combat neurodegeneration.
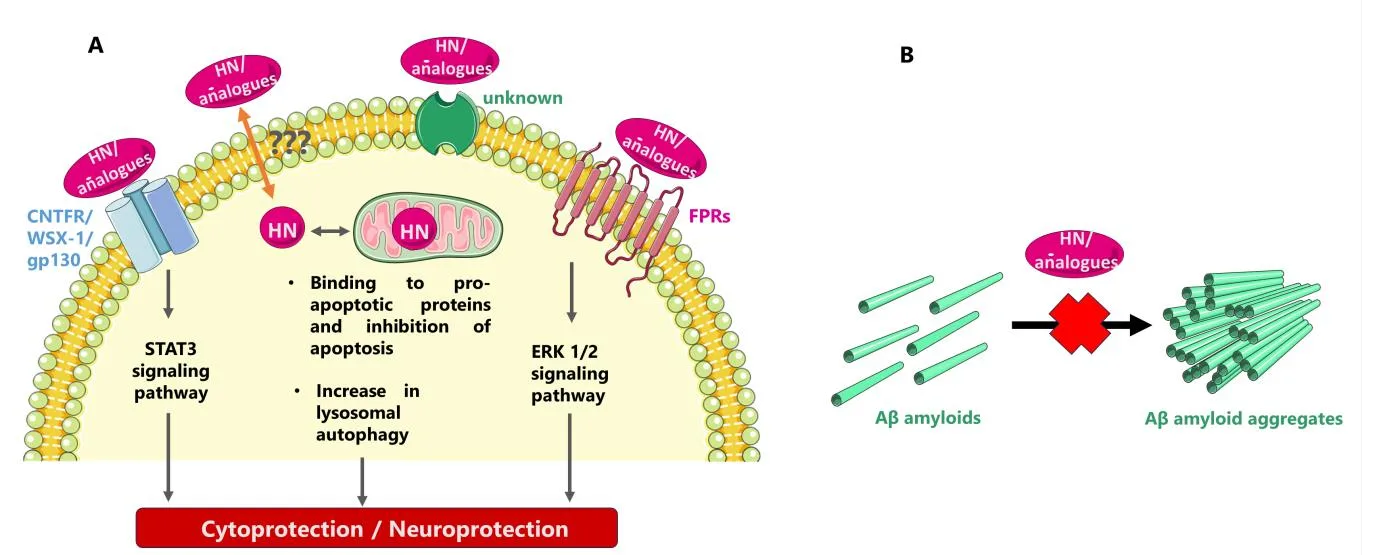
Intracellular Protection
Within the cell, Humanin binds to pro-apoptotic proteins like Bax and IGFBP3, preventing the chain reaction that leads to cell death. It also interacts with proteins like Bid, halting their movement to mitochondria and reducing activation of caspase enzymes that dismantle neurons under stress.
Humanin also enhances autophagy, particularly chaperone-mediated autophagy (CMA), which clears damaged or misfolded proteins—a key issue in neurodegenerative diseases.
Extracellular Signaling
On the cell surface, Humanin activates survival pathways through two receptor systems:
- The gp130/WSX-1/CNTFRα complex, which stimulates STAT3 signaling
- Formyl peptide receptors (FPRs), which engage ERK1/2 pathways
This dual activation promotes resilience against oxidative damage, inflammation, and mitochondrial dysfunction.
Humanin also competes with amyloid-beta for FPR binding, preventing the formation of neurotoxic aggregates linked to Alzheimer’s disease.
Mitochondrial Renewal
Recent studies show that Humanin stimulates mitochondrial biogenesis via the PI3K/AKT pathway, restoring energy production and cellular vitality—especially important in aging neurons.
Altogether, Humanin functions not as a one-trick molecule, but as a broad-spectrum defender of the nervous system.
From Lab to Clinic: Can Humanin Transform Neurodegenerative Therapy?
Humanin’s therapeutic promise is no longer theoretical. In preclinical studies, it and its analogues have demonstrated benefits in a wide range of disease models:
- Alzheimer’s disease: Reduced plaque burden, restored memory, improved mitochondrial health
- Parkinson’s disease: Enhanced mitochondrial function and behavioral recovery
- Stroke and intracerebral hemorrhage: Protected against oxidative stress, improved functional outcomes
- Age-related cognitive decline: Increased healthspan in aged mice
Overcoming Delivery Challenges
Delivering peptides like Humanin into the brain is notoriously difficult due to the blood–brain barrier (BBB). To address this, researchers are testing:
- Nanoparticle carriers, such as FGFR1-targeted polymers that enhance brain penetration
- Intranasal administration, allowing direct access via the trigeminal nerve
- Fusion peptides, like tHN-C3, designed to cross cell membranes efficiently
- Stabilizing buffers, such as the MO formulation, to preserve peptide integrity in circulation
What’s Next?
Despite these advances, more work lies ahead. Humanin’s role in cancer remains controversial—some studies suggest it suppresses tumors, others say it may promote certain cancers like triple-negative breast cancer. This highlights the importance of context, specificity, and precision analogue design.
With the help of AI-guided peptide engineering, future Humanin analogues could be customized for disease-specific targets, balancing safety and efficacy.
Final Thoughts
Humanin may be small, but its potential is vast. As research continues to reveal the depth of its protective capabilities, this humble mitochondrial peptide could become a cornerstone in the fight against neurodegenerative disease.
Whether used as a standalone therapy or as part of combination treatments, Humanin and its analogues signal a new direction in brain health—one grounded in nature, refined by science, and driven by the urgent need for better solutions in aging and neurology.
 LinkPeptide
LinkPeptide