Rewiring the Brain with Peptides: The Science Behind GLP-1’s Cognitive Power
Abstract
Neurodegenerative diseases like Alzheimer’s and Parkinson’s are increasingly linked to metabolic dysfunction, especially insulin resistance in the brain. Recent research highlights the therapeutic promise of GLP-1, a gut-derived incretin peptide traditionally used to manage type 2 diabetes. GLP-1 receptor agonists exhibit powerful neuroprotective effects—reducing inflammation, oxidative stress, and neuronal death, while improving synaptic function and cognition. Preclinical studies show significant benefits in models of Alzheimer’s, Parkinson’s, stroke, and epilepsy. Early clinical trials in humans further support their potential to slow disease progression. Even more promising are dual GLP-1/GIP receptor agonists, which demonstrate superior brain penetration and efficacy in reversing neurodegeneration. These findings mark a paradigm shift: metabolic peptides are no longer confined to glucose control—they may soon become central to treating brain disorders. As peptide-based therapies evolve, they offer a new frontier of precision neurodegeneration treatment rooted in the gut-brain axis.
From Blood Sugar to Brain Fog: The Diabetes–Dementia Connection
For decades, type 2 diabetes mellitus (T2DM) was viewed solely as a metabolic disorder, primarily affecting insulin and blood glucose regulation. But in recent years, a growing body of research has uncovered a surprising—and deeply concerning—connection between T2DM and neurodegenerative diseases like Alzheimer’s and Parkinson’s disease. Individuals with T2DM face a significantly elevated risk of developing these conditions later in life, and the underlying mechanisms point to a common culprit: impaired insulin signaling in the brain.
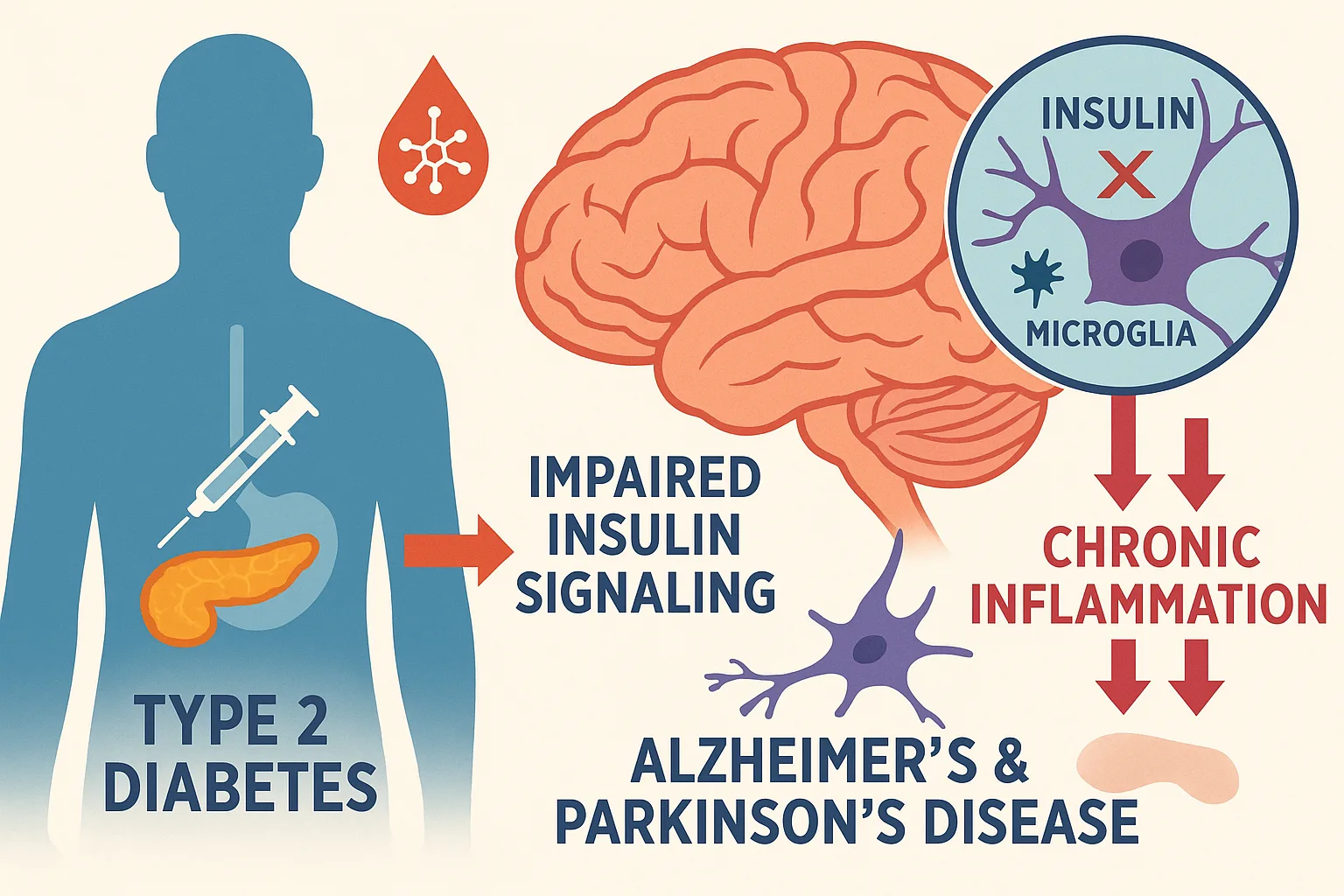
Insulin isn’t just important for managing blood sugar. In the central nervous system, it acts as a neurotrophic factor—supporting neuronal survival, synaptic plasticity, and energy metabolism. When insulin signaling becomes desensitized, neurons become less resilient, more vulnerable to oxidative stress, and less capable of repairing themselves. This dysfunction, sometimes referred to as “type 3 diabetes”, is increasingly recognized as a driver of cognitive decline.
Adding to this problem is chronic inflammation, a hallmark of both Alzheimer’s and Parkinson’s pathology. Activated microglia release pro-inflammatory cytokines that not only damage neurons but also further impair insulin signaling. It’s a vicious cycle, and one that traditional drug development has largely overlooked.
However, emerging research suggests this cycle can be broken. The application of intranasal insulin, for instance, has shown early success in improving memory and glucose metabolism in Alzheimer’s patients—without altering systemic glucose levels. These findings lay the groundwork for a new therapeutic frontier: metabolic intervention for brain disease.
GLP-1: The Gut Hormone Unlocking Brain Health
While insulin has long been the focus of metabolic research, its hormonal cousin GLP-1 (glucagon-like peptide-1) is emerging as a powerful tool in the fight against neurodegeneration. Known primarily as an incretin hormone—secreted in the gut after eating to stimulate insulin release—GLP-1 does much more than regulate blood sugar. It turns out that this 30-amino acid peptide also has remarkable effects on the brain.
GLP-1 receptors are widely expressed in neuronal tissue, especially in regions crucial for memory, motor control, and emotion, such as the hippocampus, cortex, and substantia nigra. When activated, these receptors trigger intracellular signaling cascades—including cAMP/PKA, PI3K/Akt, and MAPK pathways—that promote cell survival, mitochondrial health, synaptic plasticity, and anti-inflammatory responses.
One of GLP-1’s key advantages is that its analogues can enhance insulin sensitivity in the brain without lowering systemic glucose in non-diabetic individuals—making them safe for use in patients without T2DM. These analogues also reduce oxidative stress, inhibit apoptosis, and suppress neuroinflammation by modulating microglial activity and cytokine release.
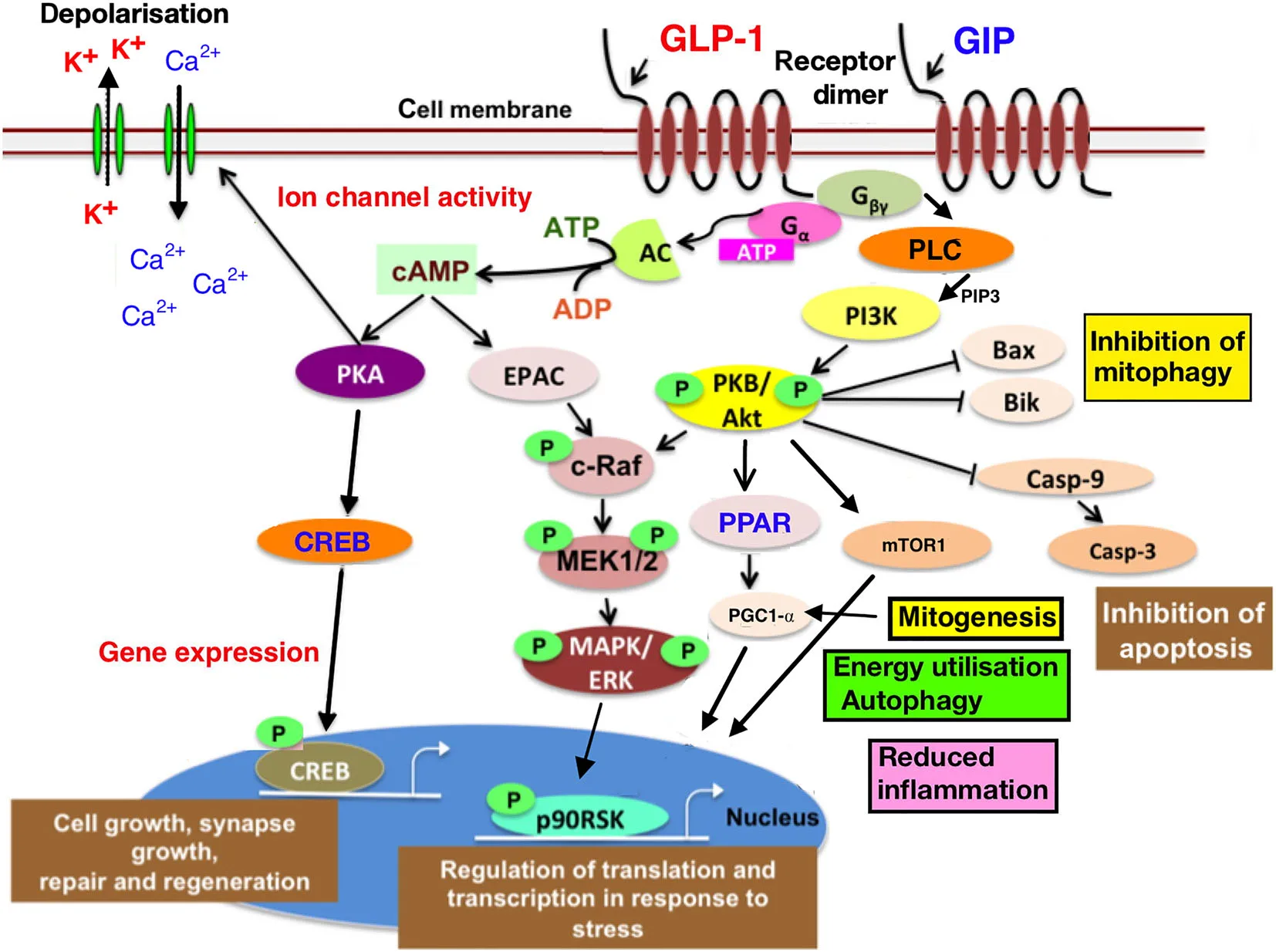
Visual evidence from animal studies supports these effects. For example, Figure 1 illustrates how GLP-1 and its sister hormone GIP activate second messenger systems that regulate energy use, gene expression, and stress resilience in neurons.
Taken together, GLP-1 is not just a metabolic hormone—it’s a neuroprotective agent with the potential to modify disease progression at the cellular level. And that has caught the attention of neurologists worldwide.
Breaking Ground: GLP-1 Agonists in Alzheimer’s and Parkinson’s Trials
The promise of GLP-1 as a neuroprotective agent has been tested extensively in preclinical studies, with consistently impressive results across models of Alzheimer’s, Parkinson’s, epilepsy, and stroke.
In Alzheimer’s disease (AD) mouse models, GLP-1 receptor agonists such as liraglutide and exendin-4 have reversed hallmark pathologies. These include reductions in amyloid plaque burden, tau hyperphosphorylation, and neuroinflammation, as well as improvements in learning, memory, and synaptic transmission. Some studies even showed that treatment started in late-stage disease still yielded cognitive benefits—suggesting disease-modifying potential, not just symptom relief.
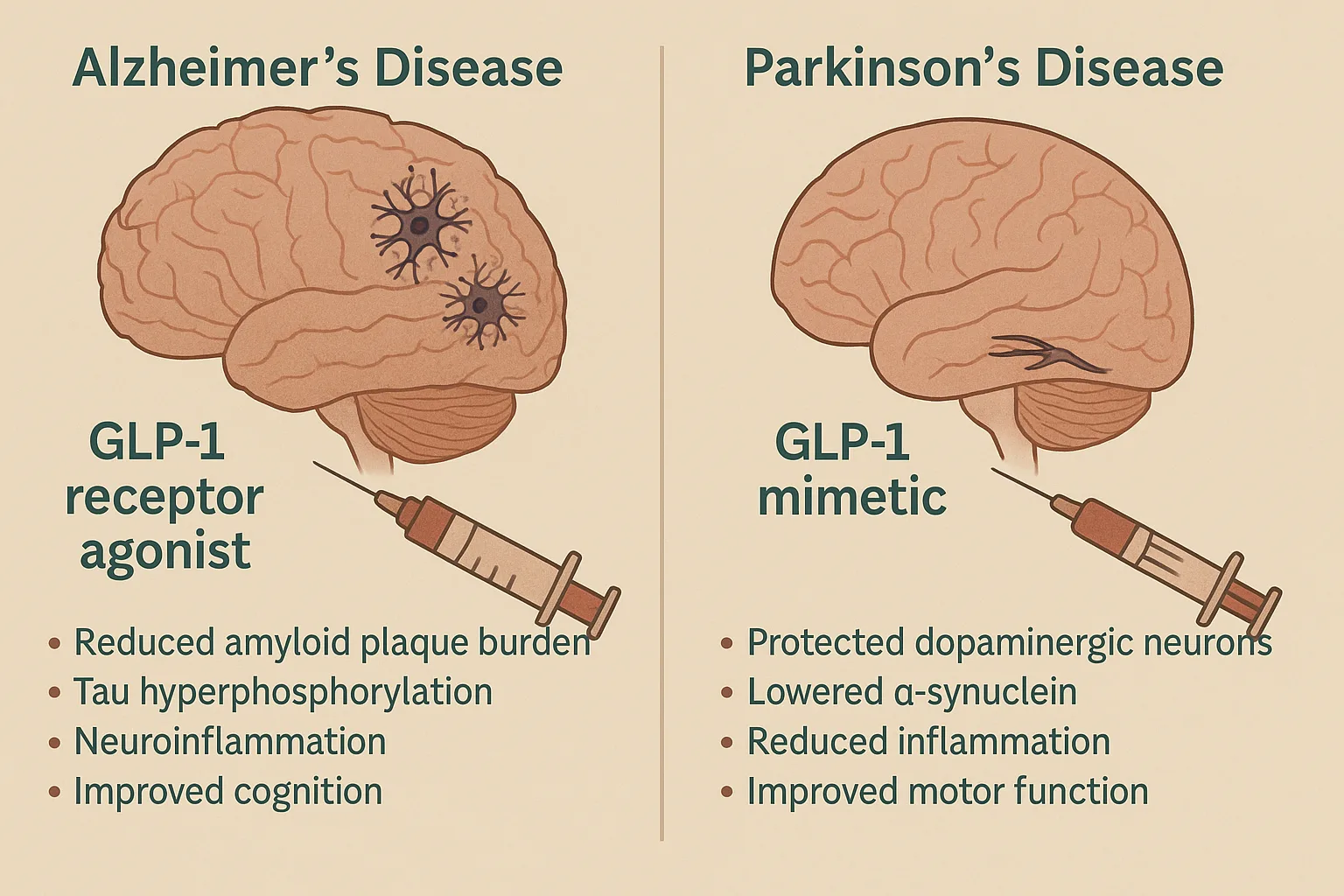
In Parkinson’s disease (PD) models, GLP-1 mimetics protect dopaminergic neurons from degeneration, especially in the substantia nigra. These drugs not only improve motor performance but also lower α-synuclein accumulation, reduce microglial activation, and normalize mitochondrial stress markers. Newer analogues like semaglutide and lixisenatide show particularly robust effects in reducing inflammation and preserving neuronal function.
The same protective trends hold in models of stroke and epilepsy. In rodent studies simulating ischemic stroke, GLP-1 drugs reduce infarct size, neuronal apoptosis, and oxidative stress, while improving neurological scores. In epilepsy models, liraglutide reduces seizure frequency, alleviates cognitive and emotional impairments, and increases levels of BDNF and anti-apoptotic proteins in the hippocampus.
But are these effects translatable to humans?
Early clinical trials suggest yes. In a landmark Parkinson’s trial, patients treated with exenatide (a GLP-1 mimetic) for 12 months showed improvements in motor scores and cognitive function—effects that persisted even after treatment ended. This durability indicates true disease modification, not just symptom suppression.
Similarly, in Alzheimer’s trials, patients receiving liraglutide maintained brain glucose metabolism as shown by FDG-PET scans, while placebo groups declined. A larger study (ELAD trial) revealed that liraglutide reduced gray matter loss and preserved executive function scores over 12 months of treatment.
These results confirm what preclinical data had long hinted: GLP-1 receptor agonists can slow or halt neurodegenerative progression in humans. While not yet mainstream therapy, these drugs represent a new class of brain-targeted treatments grounded in metabolic regulation and cellular resilience.
Double Impact: How Dual GLP-1/GIP Agonists Raise the Bar
As promising as GLP-1 receptor agonists are, scientists have taken the concept a step further by developing dual receptor agonists that target both GLP-1 and GIP receptors. GIP (glucose-dependent insulinotropic polypeptide) is another incretin hormone with overlapping and complementary roles to GLP-1. When activated together, these receptors can produce synergistic effects in the brain—enhancing neuroprotection, anti-inflammatory responses, and synaptic repair.
One of the major challenges in neurotherapeutics is crossing the blood–brain barrier (BBB). That’s where new dual agonists like DA4-JC and DA5-CH stand out. Engineered with cell-penetrating modifications, these compounds show superior brain uptake compared to traditional GLP-1 drugs like liraglutide or semaglutide. In animal studies, they offer greater protection for dopaminergic neurons, reduced α-synuclein and amyloid load, and more robust behavioral improvements.
In direct comparisons, dual agonists consistently outperform single-receptor treatments across models of Alzheimer’s, Parkinson’s, stroke, and epilepsy. These advances mark a shift from simply adapting diabetes drugs to designing CNS-specific peptides—a new generation of therapies with enhanced potency and precision.
If GLP-1 analogues opened the door to metabolic neurotherapy, dual agonists are pushing it wide open.
The Future Is Metabolic: Reimagining Brain Therapies
The emerging science around GLP-1 and GIP signaling is revolutionizing how we understand and treat neurodegenerative diseases. By targeting insulin resistance, inflammation, and neuronal energy failure, these hormones do more than manage glucose—they repair the brain from within. With both clinical and preclinical evidence pointing to true disease modification, metabolic peptide therapy offers a new path forward. And with next-generation dual agonists showing enhanced brain penetration and potency, the future of Alzheimer’s and Parkinson’s treatment may lie not just in neurology—but in endocrinology. The gut-brain axis is no longer just a theory—it’s becoming a therapeutic reality.
 LinkPeptide
LinkPeptide