The Peptide Revival: How Next-Gen Therapeutics Are Reshaping Modern Medicine
Abstract
Peptides are experiencing a scientific and commercial renaissance, emerging as a powerful class of therapeutics that bridge the gap between small molecules and biologics. Once limited by poor stability and delivery challenges, recent advances in synthetic chemistry, drug delivery technologies, and high-throughput screening platforms have transformed their potential. This blog explores the evolution of peptide drug development, from early hormone analogues and recombinant insulin to venom-derived therapeutics and macrocyclic peptides. It highlights innovative strategies to overcome pharmacokinetic barriers, such as PEGylation, lipidation, and oral delivery systems like semaglutide. Cutting-edge discovery tools—including phage and mRNA display—are enabling the rapid identification of stable, selective, and potent peptide candidates. As peptides become increasingly programmable and patient-friendly, their role in treating metabolic disorders, cancer, and immune-related conditions is expanding rapidly. With these breakthroughs, peptide therapeutics are poised to play a central role in the future of precision medicine.
The Peptide Comeback: Why This Drug Class Is Hot Again
Peptides, long considered the underdogs of drug development, are undergoing a remarkable revival. Nearly a century has passed since the clinical debut of insulin in 1922—a milestone that marked the dawn of peptide therapeutics. Since then, over 80 peptide-based drugs have reached the market, offering treatment for conditions ranging from diabetes and cancer to osteoporosis and chronic pain.
What sets peptides apart is their ability to interact with biological targets with extraordinary precision. Their inherent selectivity, potency, and typically low toxicity make them uniquely suited to modulate complex physiological pathways. Yet despite their promise, peptides have historically faced major hurdles: poor oral bioavailability, rapid degradation in the body, short circulation times, and high manufacturing costs have all limited their therapeutic reach.
Today, that narrative is changing. Revolutionary advances in peptide synthesis, molecular engineering, and drug delivery technologies are dismantling these barriers. Researchers are tapping into nature’s own pharmacopoeia—mining venoms, expanding genetic codes, and leveraging display technologies—to design peptides that are more stable, more versatile, and more clinically viable than ever before.
This blog explores the forces driving the modern peptide renaissance—and how these transformative innovations are redefining the future of medicine.
From Hormones to Venoms: Key Breakthroughs in Peptide Therapeutics
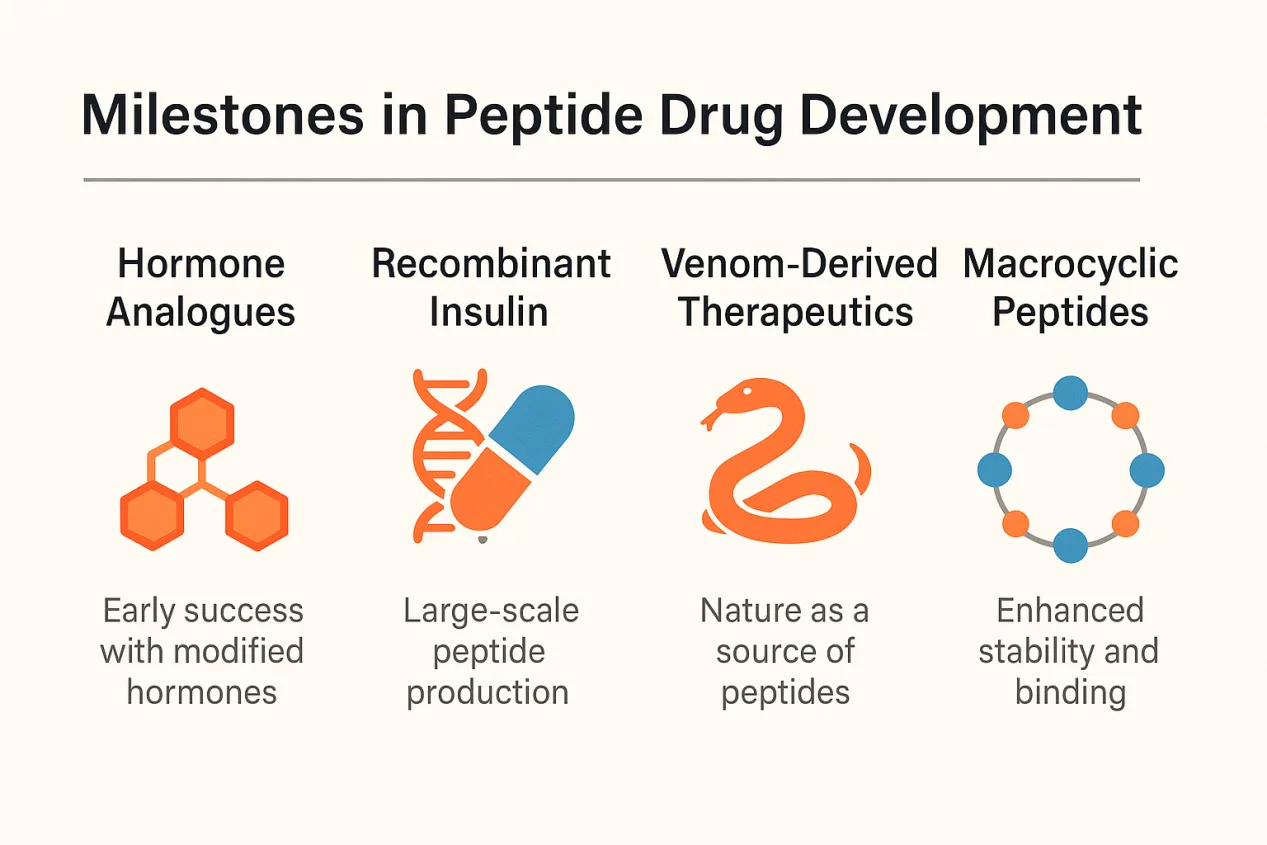
The journey of peptide drugs is marked by periods of intense innovation, strategic breakthroughs, and renewed scientific interest. After the initial success of insulin, progress in peptide therapeutics accelerated during the mid-20th century with the introduction of synthetically produced hormones such as oxytocin and vasopressin. These early developments laid the foundation for modern peptide medicine, though limitations in stability and synthesis kept progress slow.
Everything changed with the invention of solid-phase peptide synthesis (SPPS) in 1963. This breakthrough drastically reduced the time and complexity of peptide production, enabling the pharmaceutical industry to explore peptide drugs more seriously. Medicinal chemists began modifying natural peptides to improve their metabolic stability, receptor selectivity, and pharmacokinetic properties. These efforts led to clinically successful analogues like desmopressin, octreotide, and leuprolide, many of which remain in use today.
The 1980s ushered in a new era with the advent of recombinant DNA technology. This enabled large-scale production of human peptides like insulin, calcitonin, and parathyroid hormone (PTH) in microbial systems, eliminating the need for animal-derived extracts. Recombinant techniques also opened the door for engineering peptide analogues with enhanced therapeutic profiles.
Nature itself has proven to be an abundant source of inspiration. Venom peptides from animals such as cone snails, spiders, and Gila monsters have yielded unique therapeutics with exceptional potency and specificity. For example, exenatide, a GLP-1 receptor agonist from Gila monster venom, revolutionized the treatment of type 2 diabetes. Ziconotide, derived from cone snail venom, provides potent pain relief far beyond the capabilities of morphine—without the addictive side effects.
From hormones to venoms to engineered analogues, these milestones showcase the versatility of peptides and the expanding toolbox for turning them into safe, effective medicines.
Breaking Barriers: How Peptides Are Becoming Smarter and Stronger
While peptides offer compelling therapeutic advantages, their clinical development has long been hindered by a core set of challenges: rapid enzymatic degradation, poor membrane permeability, and fast renal clearance. These factors lead to short half-lives and low bioavailability, often requiring frequent injections—an inconvenience that has limited patient compliance and commercial viability.
To combat these limitations, researchers have employed a suite of chemical modifications aimed at enhancing peptide stability and pharmacokinetics. PEGylation, the attachment of polyethylene glycol chains, is a widely used strategy to shield peptides from proteolytic enzymes and prolong their circulation time. Other methods include lipidation—as seen in drugs like liraglutide and semaglutide—which binds peptides to albumin and delays their renal clearance. Fusion to larger proteins, such as human serum albumin or immunoglobulin fragments, offers another route to extend half-life and reduce dosing frequency.
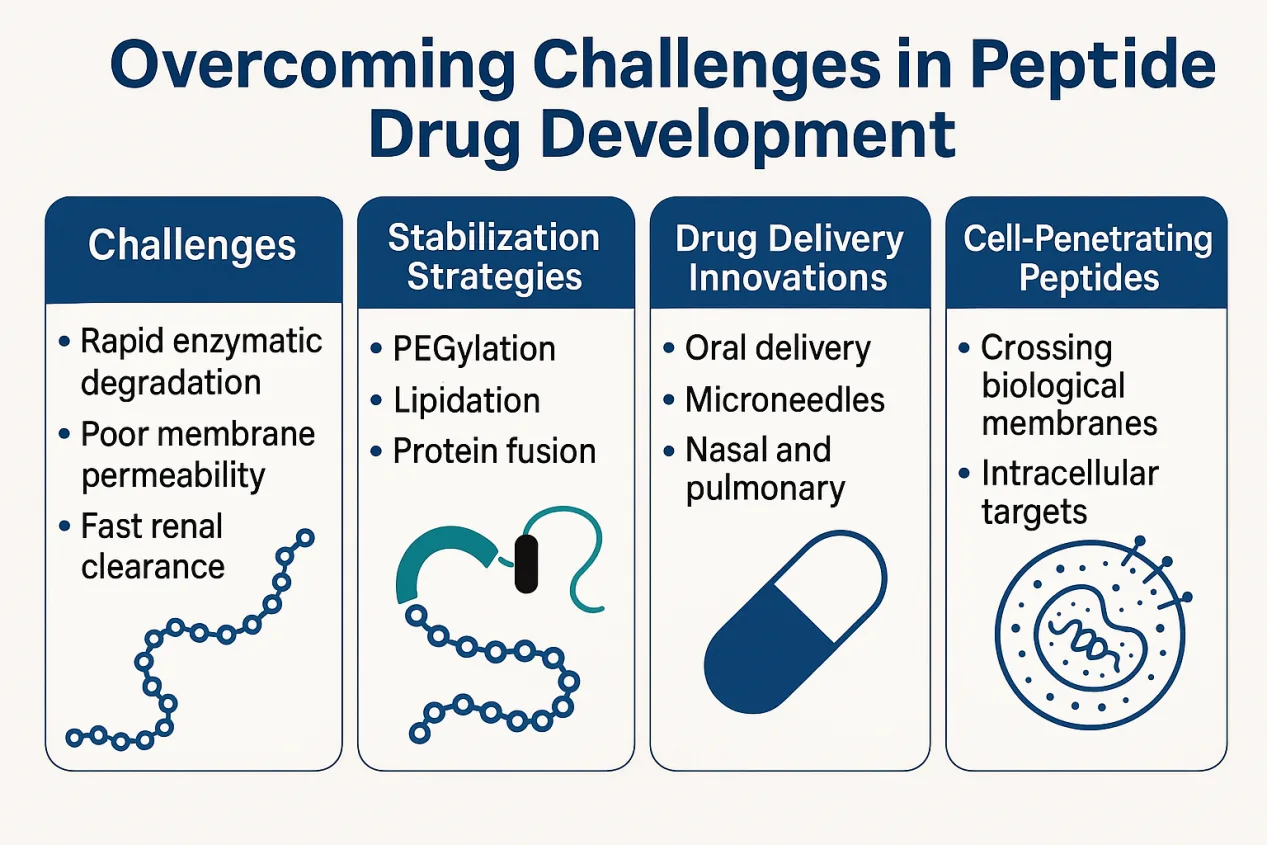
Beyond chemistry, advances in drug delivery systems are transforming how peptides reach their targets. The success of oral semaglutide, made possible by the absorption enhancer SNAC, represents a major breakthrough in non-injectable peptide delivery. Other formats, including transdermal microneedles, intranasal sprays, and pulmonary inhalation, are under development to further expand administration options.
A particularly exciting frontier lies in cell-penetrating peptides (CPPs) and stapled peptides, which have been engineered to cross biological membranes and act on intracellular targets. Compounds like nerinetide, designed for stroke therapy, and AM-111, for acute hearing loss, are advancing through clinical trials—signaling that the traditional barrier between peptides and the central nervous system may finally be crumbling.
Through a combination of smart chemistry and novel delivery technologies, peptides are shedding their old limitations and becoming more versatile, patient-friendly, and clinically powerful than ever before.
Next-Gen Discovery: The Technologies Powering Peptide Innovation
The future of peptide drug discovery is being shaped not just by chemistry, but by powerful biological tools that allow scientists to search vast molecular libraries for high-potential therapeutic candidates. Among the most transformative of these tools are display technologies, which enable the rapid identification of peptides that bind tightly and selectively to disease-relevant targets.
Phage display, one of the earliest techniques, uses bacteriophages to present billions of peptide variants on their surfaces. Through iterative rounds of selection, researchers can isolate peptides with optimal binding properties. This approach has already yielded several FDA-approved drugs, including romiplostim, a thrombopoietin receptor agonist for chronic immune thrombocytopenia, and ecallantide, a kallikrein inhibitor for hereditary angioedema.
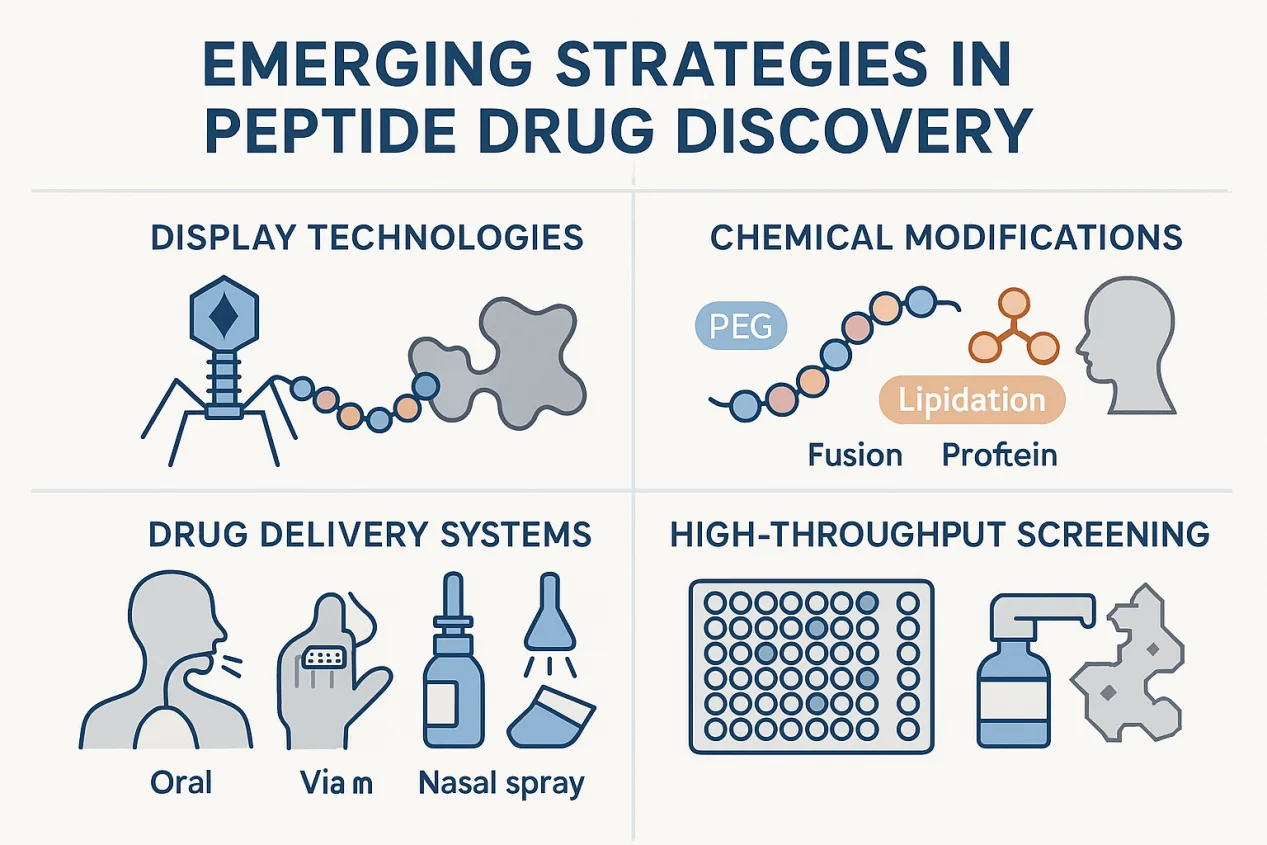
More recent platforms like mRNA display and ribosome display allow for even larger libraries and more diverse peptide scaffolds. One standout is the RaPID system, which combines mRNA display with genetic code reprogramming to incorporate non-canonical amino acids. This enables the discovery of macrocyclic peptides with enhanced stability and novel mechanisms of action.
An especially innovative technique is mirror-image phage display, which creates D-enantiomeric peptides—resistant to proteolysis and highly suitable for in vivo applications. These synthetic peptides can mimic the structure of natural L-peptides while dramatically improving pharmacological properties.
By integrating high-throughput screening with synthetic biology and unnatural amino acid chemistry, modern discovery platforms are redefining what peptides can be and how fast they can be developed—pushing the boundaries of what’s possible in drug design.
The Road Ahead: Why Peptides Are Shaping the Future of Medicine
As peptide science advances, it’s becoming clear that these molecules fill a crucial space between small-molecule drugs and large biologics. Peptides offer a rare combination of high target specificity, low toxicity, and structural adaptability, making them ideal for therapeutic targets that are difficult to modulate with traditional approaches—particularly extracellular receptors, receptor subtypes, and protein–protein interactions.
Therapeutic areas where peptides are poised to dominate include metabolic and endocrine disorders, cardiovascular disease, and oncology. Peptide-based GLP-1 receptor agonists have already transformed the treatment of type 2 diabetes and obesity, and next-generation analogues continue to show promise for long-term metabolic health. In oncology, peptides are being used not only to deliver cytotoxic agents directly to tumor cells but also in peptide receptor radionuclide therapy (PRRT) and personalized cancer vaccines.
Beyond systemic therapies, peptides are increasingly being explored for topical and localized applications, such as antimicrobial wound treatments, anti-aging skin peptides, and gut-targeted immunomodulators. Their ability to act locally with minimal systemic exposure offers a major advantage in terms of both safety and efficacy.
Looking forward, the integration of peptide discovery pipelines with computational drug design, machine learning, and automated synthesis platforms will further accelerate development timelines and lower production costs. The peptide landscape is expanding from niche biologics into a mainstream drug modality—one that’s equipped to tackle many of medicine’s most stubborn challenges.
In the era of precision medicine, peptides are no longer an alternative—they are a frontier.
Reference
Muttenthaler, M., King, G. F., Adams, D. J., & Alewood, P. F. (2021). Trends in peptide drug discovery. Nature reviews Drug discovery, 20(4), 309-325.
https://doi.org/10.1038/s41573-020-00135-8
Merrifield, R. B. (1963). Solid phase peptide synthesis. I. The synthesis of a tetrapeptide. Journal of the American Chemical Society, 85(14), 2149-2154.
https://doi.org/10.1021/ja00897a025
Luther, A., Bisang, C., & Obrecht, D. (2018). Advances in macrocyclic peptide-based antibiotics. Bioorganic & Medicinal Chemistry, 26(10), 2850-2858.
https://doi.org/10.1016/j.bmc.2017.08.006
Zhang, J., Desale, S. S., & Bronich, T. K. (2015). Polymer-based vehicles for therapeutic peptide delivery. Therapeutic Delivery, 6(11), 1279-1296.
 LinkPeptide
LinkPeptide