Serelaxin: A Novel Therapeutic Agent in Cardiovascular and Renal Disease Management
Abstract
Serelaxin, a recombinant form of human relaxin-2, has emerged as a promising therapeutic agent in the management of cardiovascular and renal diseases. Acting through the RXFP1 receptor, Serelaxin induces systemic vasodilation, reduces pulmonary and renal pressures, and offers organ protection, particularly in patients with acute heart failure (AHF). Its ability to modulate key pathways, such as endothelin-1 and angiotensin II, positions Serelaxin as a multifaceted therapeutic with potential applications beyond AHF, including chronic heart failure, coronary artery disease, and renal impairment. Clinical trials like RELAX-AHF-1 and RELAX-AHF-2 have demonstrated significant reductions in mortality and renal deterioration, though challenges in fibrosis-related conditions highlight the complexity of its therapeutic use. This review explores Serelaxin’s mechanisms, clinical benefits, and future potential, offering insight into its expanding role in modern pharmacology.
Serelaxin – A Breakthrough in Cardiovascular and Renal Therapeutics
Serelaxin, a recombinant form of human relaxin-2, has gained attention as a therapeutic agent with significant potential in the management of cardiovascular and renal disorders. Relaxin-2 is a naturally occurring peptide hormone, primarily recognized for its role during pregnancy, but research has revealed its broader biological functions. Serelaxin binds to the relaxin family peptide receptor 1 (RXFP1), triggering vasodilation, enhanced renal blood flow, and modulation of fibrosis pathways, making it a promising candidate for treating acute heart failure (AHF) and other cardiovascular diseases[1].
The development of Serelaxin as a drug was motivated by its ability to reduce systemic vascular resistance and improve renal perfusion, offering organ protection. Its pharmacological properties make it particularly valuable in managing patients with AHF, where it has demonstrated reduced mortality and hospital readmission rates in clinical trials[2]. Beyond AHF, studies suggest that Serelaxin could be beneficial in treating chronic heart failure, coronary artery disease, and renal impairment, highlighting its expanding therapeutic scope[3].
As ongoing clinical trials continue to explore its potential, Serelaxin stands out as a novel therapeutic in modern medicine, offering new avenues for addressing complex cardiovascular and renal conditions.
The Science Behind Serelaxin’s Multifaceted Effects
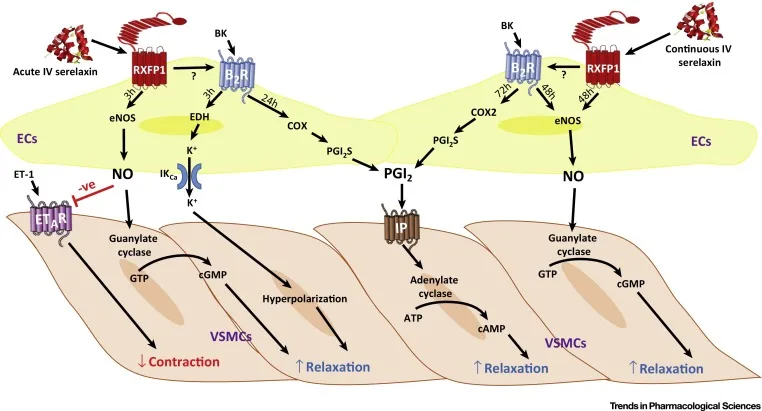
Serelaxin exerts its therapeutic effects primarily through its interaction with the relaxin family peptide receptor 1 (RXFP1). Upon binding to RXFP1, Serelaxin activates a range of intracellular signaling pathways that promote vasodilation, reduce inflammation, and modulate fibrosis. One of the key mechanisms is the stimulation of nitric oxide (NO) production, leading to vascular relaxation and enhanced renal blood flow[4]. This vasodilatory effect helps alleviate the hemodynamic stress on the heart in patients with acute heart failure (AHF) by reducing both preload and afterload.
Additionally, Serelaxin has been shown to interact with pathways involving endothelin-1 (ET-1) and angiotensin II (Ang II), two major contributors to vasoconstriction and fibrosis. By modulating these pathways, Serelaxin reduces systemic vascular resistance and improves blood flow, particularly in the kidneys and lungs[5]. In preclinical models, Serelaxin was found to increase the expression of endothelin receptor type B (ETB), which is linked to the promotion of vasodilation and reduction of vascular stiffness[6].
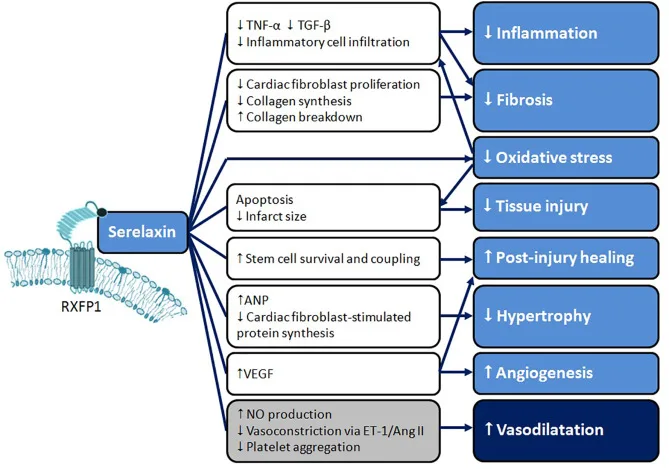
Serelaxin’s ability to influence fibrosis is another important aspect of its pharmacological action. By inhibiting the activity of transforming growth factor-beta (TGF-β), a key mediator of fibrosis, Serelaxin has shown potential in preventing the progression of fibrotic diseases, including heart and kidney fibrosis[6]. This combination of vasodilation and anti-fibrotic properties makes Serelaxin a valuable therapeutic agent in cardiovascular and renal diseases.
Validating Serelaxin’s Efficacy in Cardiovascular Care
Clinical trials have played a pivotal role in establishing the therapeutic potential of Serelaxin, particularly in the management of acute heart failure (AHF). One of the most notable trials is the Phase III RELAX-AHF-1 study, which demonstrated that Serelaxin significantly reduced mortality rates and hospitalization in AHF patients. The study found that patients treated with Serelaxin showed marked improvements in renal function, as indicated by increased glomerular filtration rates and reduced serum creatinine levels[7]. These findings highlight Serelaxin’s unique ability to protect vital organs such as the heart and kidneys, both of which are critically affected in AHF.
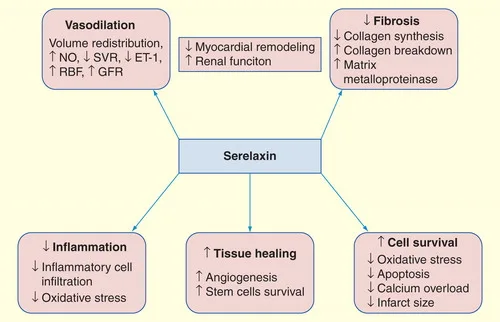
In addition to its efficacy in AHF, Serelaxin has shown potential in treating chronic heart failure (HF) and conditions associated with systemic fibrosis. Clinical studies suggest that Serelaxin’s anti-fibrotic properties, particularly its inhibition of the transforming growth factor-beta (TGF-β) pathway, could be beneficial for patients with chronic heart failure and those at risk for fibrosis-related complications[2].
The RELAX-AHF-2 study, which is currently ongoing, aims to further validate these findings by assessing the long-term effects of Serelaxin on cardiovascular mortality and heart failure outcomes. Preliminary data from these trials suggest that Serelaxin may become a key therapeutic agent for managing cardiovascular diseases beyond AHF. As clinical research advances, the role of Serelaxin in treating renal dysfunction and other conditions associated with endothelial damage and fibrosis is expected to expand.
Addressing Limitations and Optimizing Serelaxin’s Use
While the potential of Serelaxin as a therapeutic agent in cardiovascular and renal diseases is substantial, there are notable challenges that require careful consideration. One of the key hurdles is the variability in patient responses, particularly in conditions associated with fibrosis. For instance, in systemic sclerosis (SSc), where fibrosis is a hallmark feature, clinical trials with Serelaxin did not yield the expected outcomes. Despite its proven anti-fibrotic effects in preclinical models, human trials, such as the study by Khanna et al. (2009), failed to demonstrate significant improvements in skin fibrosis using the Modified Rodnan Skin Score (MRSS), the standard measure of fibrotic involvement in SSc[2,3].
Another challenge relates to species-specific differences in the physiological role of relaxin. While animal studies have shown strong vasodilatory and anti-fibrotic effects, these results have not always translated directly to human subjects. Differences in relaxin signaling pathways between species, especially regarding the expression and activity of RXFP1 receptors, contribute to the complexity of optimizing Serelaxin’s therapeutic efficacy.
Safety and immunogenicity also remain areas of focus. Since Serelaxin is a recombinant form of a naturally occurring hormone, the development of neutralizing antibodies in patients undergoing repeated dosing could pose a risk. However, results from studies like the RELAX-REPEAT trial suggest that antibody formation is rare and has not been associated with adverse clinical outcomes. Addressing these challenges is critical to maximizing Serelaxin’s clinical utility.
The Future of Serelaxin in Advanced Medical Treatment
Serelaxin, a recombinant form of the human relaxin-2 hormone, has shown significant promise in the treatment of acute heart failure (AHF) and other cardiovascular conditions. Acting through the RXFP1 receptor, it exerts potent vasodilatory effects by increasing nitric oxide (NO) production and modulating key pathways like endothelin-1 (ET-1) and angiotensin II (Ang II). Its ability to improve renal blood flow and reduce systemic vascular resistance offers valuable organ protection, particularly for patients with AHF. Clinical trials, such as RELAX-AHF-1, have demonstrated Serelaxin’s potential to reduce mortality and prevent worsening renal function.
Despite these successes, challenges remain in expanding Serelaxin’s therapeutic applications, particularly in fibrosis-related conditions like systemic sclerosis (SSc), where clinical outcomes were less favorable. Additionally, the translational challenges between preclinical models and human trials underscore the complexity of optimizing Serelaxin’s therapeutic benefits. Issues related to immunogenicity, while largely resolved, still require attention, especially for long-term administration.
Looking forward, Serelaxin’s role in managing chronic heart failure, coronary artery disease, and fibrotic conditions will depend on overcoming these challenges. Continued research is essential to fully unlock Serelaxin’s potential as a multifaceted therapeutic agent in cardiovascular and renal diseases, ultimately improving patient outcomes and broadening its clinical utility.
 LinkPeptide
LinkPeptide