Smart Healing: How Peptide-Based Coatings Are Revolutionizing Bone Implants
Abstract
Orthopedic implant success hinges on effective osseointegration, which can be disrupted by early-stage inflammation and oxidative stress. This study introduces a novel peptide-based, inflammation-responsive coating designed to sequentially regulate immune response and promote bone regeneration. The coating consists of a self-assembling peptide hydrogel incorporating anti-inflammatory peptides linked via ROS-cleavable bonds and osteogenic peptides to support later-stage healing. Upon exposure to reactive oxygen species (ROS), commonly elevated during post-surgical inflammation, the coating releases anti-inflammatory agents to calm the immune environment. As inflammation subsides, the remaining scaffold promotes osteogenic differentiation of stem cells. In vitro experiments demonstrated reduced oxidative stress and enhanced bone-related gene expression, while in vivo studies in rat models confirmed improved osseointegration and bone-implant bonding. This dual-function system represents a significant advancement in biomaterial design, offering a smart, bioadaptive strategy for enhancing the healing and longevity of orthopedic implants by aligning material performance with the body’s natural healing phases.
Introduction: When Healing Fails to Connect
Orthopedic implants have transformed modern medicine, offering hope and mobility to patients with bone injuries, degenerative conditions, or those undergoing joint replacements. Yet, despite advances in surgical techniques and biomaterials, one persistent problem remains: osseointegration, or how well the implant bonds with the surrounding bone.
A major reason for poor integration is inflammation—the body’s immediate and natural response to injury or surgical trauma. While essential for kickstarting healing, excessive or prolonged inflammation can actually hinder the regenerative process, leading to implant failure over time.
But what if we could give implants the intelligence to respond to their environment—calming inflammation when it’s high and encouraging regeneration when it’s time to heal?
That’s exactly what a recent study proposes with a breakthrough in peptide-based, inflammation-responsive implant coatings. Let’s take a closer look at how this innovation works and why it could be a game changer for orthopedic care.
The Hidden Barrier: Inflammation at the Bone-Implant Interface
The moment an implant enters the body, it disrupts tissue and triggers a wave of immune activity. Macrophages, a type of immune cell, rush to the site, releasing pro-inflammatory signals like cytokines and reactive oxygen species (ROS). These signals are useful at first—they help clean the site and prevent infection—but too much ROS can create an oxidative environment that damages tissue, delays healing, and impairs bone growth.
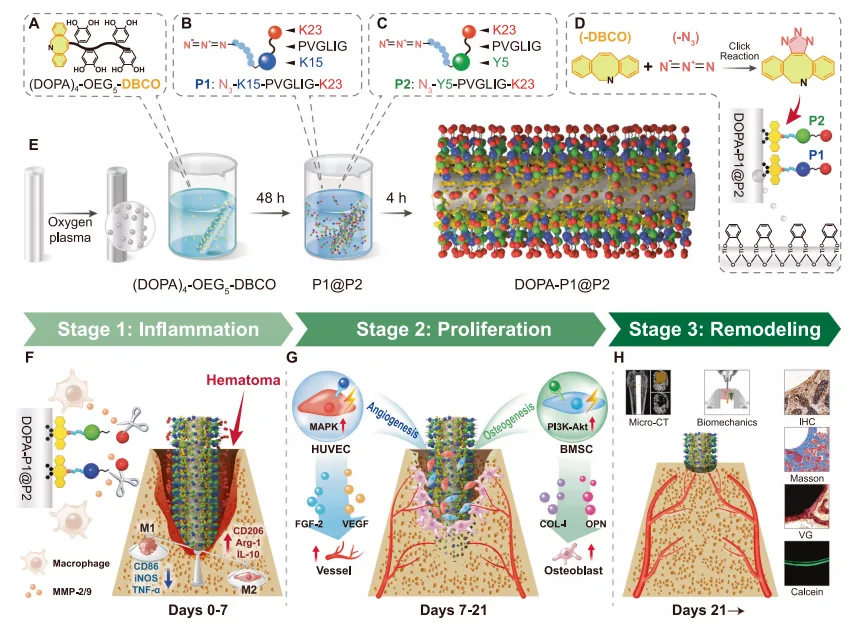
The synthesis of DOPA-P1@P2 and its promotion of osseointegration via
temporal regulation of bone regeneration.
Traditionally, implants are coated with bioinert or bioactive materials meant to passively support bone bonding. However, these coatings do not account for the highly dynamic biological conditions in the early healing window. This mismatch between implant design and biological reality has inspired researchers to seek smarter, more adaptive solutions.
A Smarter Solution: The Power of Responsive Peptides
The core idea of this study is to design an implant coating that responds to the body’s inflammatory cues—essentially, a material that “knows” when to fight inflammation and when to switch to promoting regeneration.
Here’s how it works:
Peptide Hydrogel Foundation
The coating is made from a self-assembling peptide hydrogel, which serves as a scaffold around the implant. Peptides—short chains of amino acids—can be customized for specific biological functions, such as reducing inflammation or stimulating cell growth.
ROS-Cleavable Linker
Embedded in this hydrogel are anti-inflammatory peptides, which are attached via a clever linker that is sensitive to ROS. When ROS levels spike (as they do during inflammation), the linkers are cleaved, and the anti-inflammatory peptides are released.
Sequential Action
After the inflammatory response subsides, the remaining peptide matrix shifts gears. It provides osteogenic (bone-forming) support, encouraging the migration, adhesion, and differentiation of mesenchymal stem cells (MSCs) into bone-building osteoblasts.
In other words, the coating senses the body’s needs and delivers the right therapeutic action at the right time.
How It Works: A Two-Phase Healing Strategy
To validate their design, the researchers conducted both in vitro (lab-based) and in vivo (animal) experiments.
In vitro results:
The coating effectively reduced ROS levels and inflammatory markers in immune cells exposed to oxidative stress.
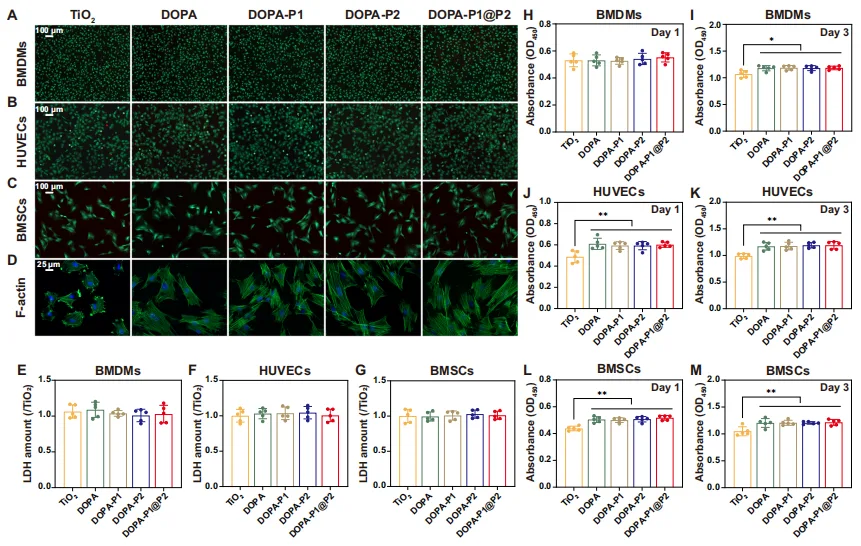
Cytocompatibility assessment of modified surfaces.
MSCs grown on the coating showed increased activity of osteogenic genes, such as RUNX2, ALP, and OCN, which are key markers of bone formation.
The coating also encouraged better cell adhesion and proliferation, indicating a healthy and supportive environment for tissue growth.
In vivo results:
In rat models with implanted titanium rods (common in orthopedic implants), the coated implants demonstrated stronger and more stable bone integration than uncoated or traditionally coated implants.
Micro-CT imaging and histological analysis confirmed higher bone volume and better bone-implant contact in the coated group.
Importantly, inflammatory markers were lower, showing that the coating had a clear modulatory effect on the immune environment.
Why It Matters: Real Benefits for Real Patients
These findings may sound technical, but the implications are far-reaching—especially for patients facing joint replacements, spinal surgeries, or complex fractures.
Here’s why this matters:
Fewer Complications: By reducing inflammation at the source, these coatings could lower the risk of post-operative complications such as chronic pain, implant loosening, or infection.
Faster Healing: A healthier local environment supports quicker and more complete bone regeneration, shortening recovery time and improving mobility.
Longer-Lasting Implants: Stronger osseointegration means implants are less likely to fail or need revision surgeries, which are costly and traumatic for patients.
In essence, this peptide coating bridges the gap between immunology and regeneration, providing a sequential, biomimetic approach to healing.
The Science of Peptides: Small Molecules, Big Impact
What makes peptides so special for this application?
Peptides are small, customizable, and highly bioactive molecules. They can mimic signals found naturally in the body, making them ideal for influencing cell behavior. In this coating system:
Anti-inflammatory peptides were selected to reduce macrophage activation and cytokine release.
Osteogenic peptides helped promote MSC differentiation and matrix mineralization.
The ROS-sensitive linker ensured these biofunctions were activated only when needed, providing a time-sensitive, controlled therapeutic effect.
This elegant design offers precision without the complexity or risk of drug-based therapies, which often suffer from side effects or uncontrolled release profiles.
Beyond Bone: A Platform for Responsive Biomaterials
While this study focused on titanium implants in bone tissue, the concept of environmentally responsive peptide coatings has potential far beyond orthopedics.
Future applications could include:
Dental implants, which also require strong bone integration.
Neural implants, where inflammation can impair performance.
Wound dressings that adapt to infection or oxidative stress.
Tissue engineering scaffolds for organs and joints.
With the rising interest in regenerative medicine and personalized biomaterials, this approach could be adapted and scaled for a wide variety of clinical needs.
Final Take: Healing, Reimagined
The idea of implants that think—or at least respond intelligently to the body—is no longer a distant dream. This study’s peptide-based, inflammation-responsive coating is a clear example of how materials science and biology can come together to create next-generation medical devices.
Instead of passively existing in the body, future implants will likely be active participants in healing—sensing, responding, and guiding tissue regeneration like never before.
For clinicians, patients, and researchers alike, the promise is clear: better healing, stronger outcomes, and a smarter path forward for implant technology.
 LinkPeptide
LinkPeptide