The Hidden Signals: Leptin, Ghrelin, and the Battle for Balance
Abstract
This blog explores the critical roles of leptin and ghrelin — two key hormones that regulate appetite and energy balance — and how their dysfunction contributes to obesity. Leptin, produced by fat cells, signals satiety, while ghrelin, secreted by the stomach, stimulates hunger. In individuals with obesity, leptin resistance prevents the brain from recognizing fullness despite high leptin levels, and ghrelin resistance disrupts normal hunger cues. The hormonal imbalance between these two signals makes weight regulation more challenging and often leads to overeating. Genetic variations, diet, and lifestyle also influence how these hormones function. Understanding this hormonal interplay shifts the perspective of obesity from being purely behavioral to being deeply biological. By addressing these underlying mechanisms, future treatments may become more targeted, effective, and sustainable. This blog offers insights into the science of appetite regulation and highlights the importance of hormonal balance in managing obesity.
Why Understanding Hunger Hormones Is Key to Tackling Obesity
Obesity is a growing global health crisis, affecting more than 650 million adults worldwide and contributing to a wide range of chronic diseases. While diet and physical activity remain central to weight management, scientists have increasingly turned their attention to the hormonal mechanisms that influence appetite and energy balance.
Two of the most critical hormones involved in this process are leptin and ghrelin. These chemical messengers help regulate when we feel hungry and when we feel full. Leptin, produced by fat cells, signals satiety to the brain and plays a key role in reducing food intake. In contrast, ghrelin is released primarily by the stomach and stimulates hunger, preparing the body for food consumption.
In individuals with a healthy weight, leptin and ghrelin function in a feedback loop that helps maintain energy balance. However, in people with obesity, this system often malfunctions. Elevated levels of leptin are commonly observed, yet the body fails to respond to its appetite-suppressing effects – a condition known as leptin resistance. At the same time, ghrelin resistance may occur, where ghrelin levels are lower than expected, but the drive to eat remains strong.
This blog will explore the science behind leptin and ghrelin, their roles in appetite regulation, and how their dysregulation contributes to obesity. By understanding these hormones, we can gain deeper insight into the biological challenges of weight control and identify new avenues for treatment.
Leptin Explained: The Fullness Hormone That Stops You from Overeating
Leptin is a hormone that plays a central role in regulating energy balance by signaling satiety – the feeling of being full – to the brain. It is primarily produced by adipose tissue (fat cells), and its levels in the bloodstream are directly proportional to the amount of body fat an individual has. In other words, the more fat you have, the more leptin your body should produce.
Leptin’s primary action occurs in the hypothalamus, a region of the brain responsible for controlling hunger and metabolism. When leptin binds to receptors in the hypothalamus, it suppresses appetite and stimulates energy expenditure. This helps maintain a stable body weight over time. However, the situation becomes more complex in the context of obesity.
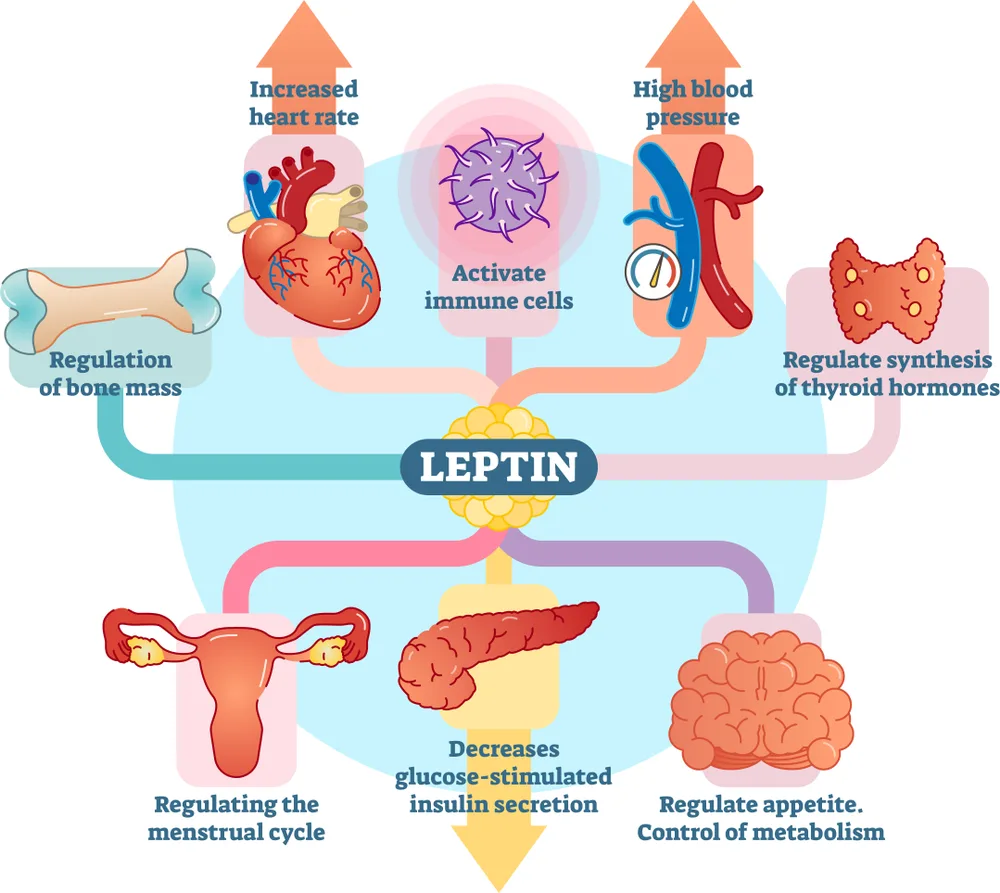
Paradoxically, many individuals with obesity have high levels of circulating leptin but do not experience its appetite-suppressing effects. This condition is known as leptin resistance – the brain becomes less responsive to leptin’s signals, leading to continued feelings of hunger despite adequate or excessive fat stores.
Leptin resistance is believed to result from impaired signaling in the brain, inflammation, and even genetic mutations. Certain rare mutations in the LEP gene (which encodes the leptin protein) or the LEPR gene (which encodes the leptin receptor) can cause severe early-onset obesity. In these cases, leptin replacement therapy has shown dramatic success. However, in more common forms of obesity, where leptin resistance is the problem, treatment remains a challenge.
Understanding leptin’s function and the mechanisms behind leptin resistance is essential to developing more effective strategies for weight management and obesity prevention.
Ghrelin: The Hunger Hormone That Keeps You Coming Back for More
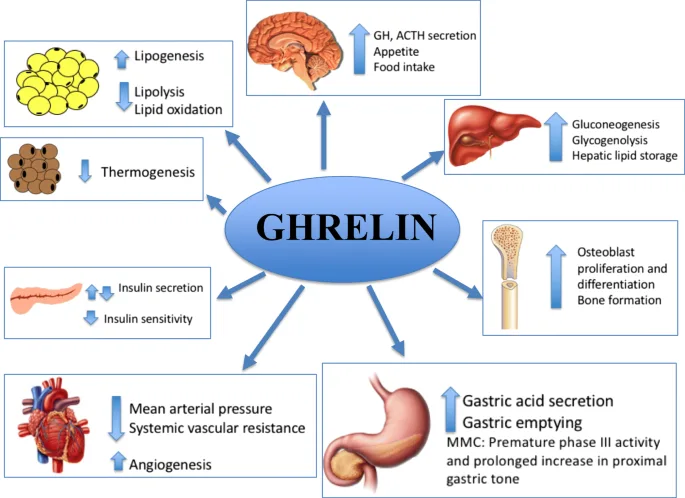
If leptin is the hormone that tells you when to stop eating, ghrelin is the one that tells you when to start. Often referred to as the “hunger hormone,” ghrelin is primarily produced by the stomach and released into the bloodstream when the stomach is empty. Its main function is to stimulate appetite and prepare the body for food intake.
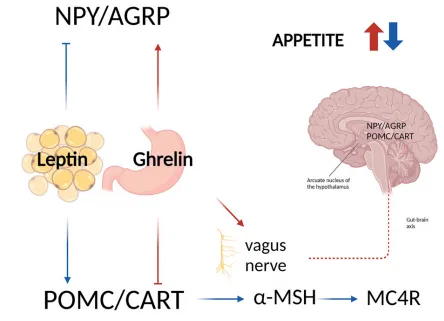
Ghrelin levels rise sharply before meals and decrease soon after eating. This natural rhythm helps regulate meal timing and food consumption. The hormone acts directly on the arcuate nucleus of the hypothalamus, where it activates neurons responsible for increasing hunger – particularly those that produce neuropeptide Y (NPY) and Agouti-related peptide (AgRP), two potent appetite-stimulating substances.
Interestingly, individuals with obesity often have lower circulating levels of ghrelin than those with a healthy body weight. This may seem counterintuitive – why would a hunger hormone be lower in people who tend to overeat? The answer may lie in what researchers refer to as ghrelin resistance. Similar to leptin resistance, this phenomenon occurs when the brain no longer responds effectively to ghrelin signals, disrupting appetite regulation and contributing to overeating.
Genetics may also play a role. Mutations and variations in the GHRL gene (which encodes ghrelin) and the GHSR gene (which encodes its receptor) have been associated with altered appetite, early-onset obesity, and even short stature in some cases. These genetic differences could affect how much ghrelin is produced or how well the body responds to it.
Ghrelin not only impacts hunger but also influences how the body stores fat, regulates insulin, and even affects mood and reward behavior. This makes it a fascinating target in obesity research – one that holds promise for both understanding and potentially treating disordered eating patterns.
When Signals Clash: How Hormonal Imbalance Fuels Obesity
Leptin and ghrelin do more than just work in isolation – they are part of a highly coordinated system that regulates hunger, satiety, and energy balance. Think of them as two opposing signals: one telling your brain you’re full (leptin), and the other telling it you’re hungry (ghrelin). The interaction between these hormones is crucial for maintaining a stable body weight.
Both hormones act on the arcuate nucleus (ARC) of the hypothalamus, targeting two key types of neurons. Leptin activates POMC and CART neurons, which reduce appetite, and inhibits NPY and AgRP neurons, which stimulate hunger. Ghrelin, on the other hand, has the opposite effect – it activates the hunger-promoting NPY/AgRP neurons and suppresses the satiety-inducing POMC/CART neurons.
In individuals with obesity, this communication becomes disrupted. The brain often becomes resistant to leptin’s “stop eating” signal, while the expected hunger response to ghrelin may persist or become dysregulated. This dual resistance creates a scenario where the brain receives confusing messages: it’s not responding to fullness, yet it continues to seek food.
Adding to the complexity, these hormones also influence reward systems in the brain. Ghrelin, for example, can increase dopamine activity, reinforcing the pleasure associated with eating – especially high-calorie, palatable foods. This hedonic drive can override normal appetite control, leading to habitual overeating even in the absence of true hunger.
Understanding this hormonal tug of war highlights why obesity isn’t simply a matter of willpower. It’s a physiological condition, rooted in a malfunctioning feedback system that makes weight loss more difficult than it appears. Addressing both leptin and ghrelin imbalances may be key to developing more effective and compassionate approaches to obesity management.
Eat Smarter: How Diet and Lifestyle Impact Your Hunger Hormones
While hormones like leptin and ghrelin play powerful roles in appetite regulation, lifestyle choices – particularly diet – can significantly influence how these hormones function. By understanding how certain nutrients and behaviors affect hormonal balance, we can make more informed decisions that support healthy weight management.
Macronutrients, for instance, impact ghrelin levels in different ways. Studies show that protein is the most effective macronutrient for suppressing ghrelin and promoting satiety, followed by carbohydrates. Fats, while essential in moderation, tend to be less effective at reducing hunger on a per-calorie basis. This means that a high-protein diet can help you feel fuller for longer, potentially leading to reduced calorie intake.
On the other hand, micronutrients such as vitamin C, zinc, and vitamin A have been shown to influence leptin levels. For example, vitamin C has been found to decrease leptin secretion in some studies, while zinc supplementation has been linked to improved leptin sensitivity in children with obesity. However, results across studies vary, and more research is needed to fully understand these effects.
Meal timing also plays a role, especially when it comes to ghrelin. Ghrelin levels naturally rise before meals and fall afterward, helping to regulate eating patterns. Eating at consistent times each day can help stabilize this rhythm, while erratic eating habits may disrupt it.
Perhaps most importantly, weight loss itself can help reset hormone sensitivity. Reducing body fat can lower leptin resistance and restore more normal ghrelin responses. However, crash dieting or overly restrictive plans can backfire, leading to spikes in ghrelin and intense hunger – which is why balanced, sustainable approaches are recommended.
In summary, while hormones influence appetite from behind the scenes, your daily choices still matter. Strategic nutrition, consistent meal timing, and sustainable weight management can all help restore a healthier hormonal environment – making it easier to reach and maintain a healthy weight.
Reframing Obesity: Why Hormonal Balance Matters More Than Willpower
Leptin and ghrelin play key roles in regulating hunger and satiety, acting as messengers between the body and brain. In people with obesity, this hormonal system often becomes disrupted. Leptin levels are typically high, but the brain resists its signal to stop eating – a condition known as leptin resistance. Meanwhile, ghrelin levels may be low, yet appetite remains strong, suggesting ghrelin resistance.
These imbalances make weight loss more difficult and highlight that obesity is not just about willpower – it’s a biological condition influenced by complex hormonal signals. By understanding how leptin and ghrelin work, we can better appreciate the challenges of managing weight and support more effective, science-backed solutions.
Continued research may pave the way for targeted therapies that restore hormonal balance and improve long-term outcomes in obesity treatment.
 LinkPeptide
LinkPeptide