Smart Sequences: How AI is Redefining the Promise of Peptides in Medicine and Skincare
Abstract
Peptides have emerged as powerful and highly specific therapeutic agents across a broad range of biomedical and cosmetic applications. Their ability to mimic natural biological functions while minimizing systemic toxicity makes them attractive candidates for targeted drug delivery, metabolic regulation, immune modulation, and advanced skincare. However, limitations in stability, delivery, and bioavailability have historically hindered their clinical translation. Recent innovations in nanocarrier systems, chemical modifications, and injectable and oral formulations are addressing these challenges. Simultaneously, the integration of artificial intelligence (AI) is revolutionizing peptide discovery, optimization, and personalization. Through deep learning, predictive modeling, and de novo sequence generation, AI accelerates the development of next-generation peptide therapeutics and cosmeceuticals. This blog explores the multifaceted roles of peptides, the evolution of delivery technologies, and the transformative synergy between computational tools and molecular science, offering insights into the future of precision medicine and intelligent skincare.
Peptides Meet AI: Reimagining Therapeutics Through Molecular Precision
Peptides are rapidly reclaiming center stage in biomedical research and therapeutic innovation. Once limited by challenges in stability, delivery, and synthesis, these short amino acid chains are now recognized as potent, highly specific agents capable of modulating complex biological pathways with minimal off-target effects. From metabolic disorders and cancer to antimicrobial resistance and dermatological applications, peptide-based interventions are expanding the frontiers of precision medicine.
Driving this resurgence is the transformative influence of artificial intelligence (AI). As peptide science enters an era of increasing molecular complexity, AI offers a means to decode, design, and deploy peptide candidates with unprecedented speed and accuracy. Machine learning models are now capable of predicting binding affinities, optimizing structural motifs, and identifying therapeutic leads far more efficiently than traditional empirical methods.
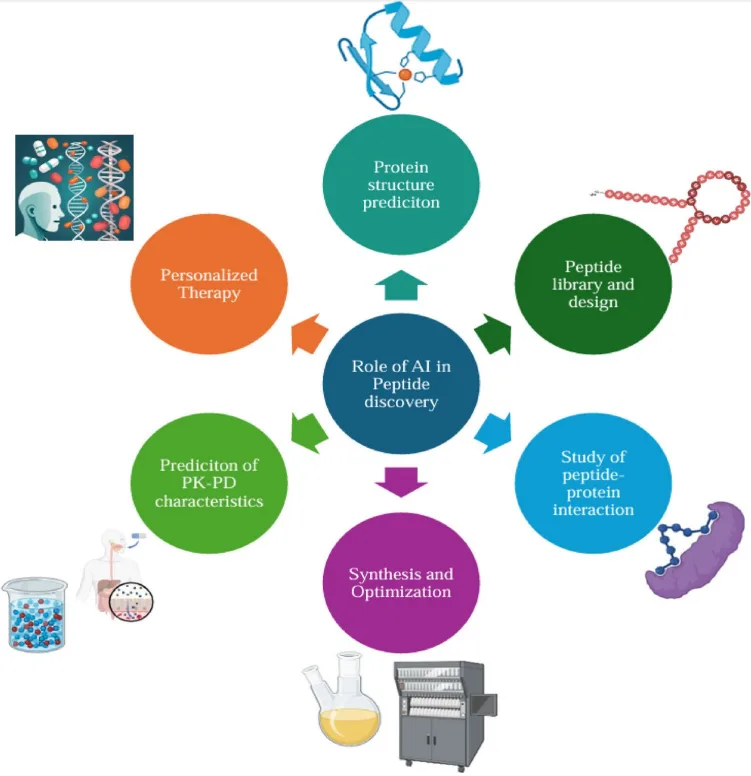
This convergence of peptide biochemistry and computational intelligence is not merely accelerating discovery—it is redefining what is possible. In this blog, we examine how AI-enhanced peptide research is enabling a new generation of therapeutics and cosmeceuticals, bridging gaps between structure, function, and translational impact across medicine and biotechnology.
From Hormones to Oncology: The Versatile Roles of Therapeutic Peptides
Peptides are no longer confined to their historical roles as hormones or signaling molecules. Advances in synthetic chemistry, molecular biology, and structural modeling have revealed their potential as multifunctional agents across therapeutic domains. Today, peptides are engineered to serve as enzyme inhibitors, antimicrobial agents, immune modulators, drug delivery vectors, and even cosmetic actives—each class characterized by unique mechanisms of action and high target specificity.
Therapeutically, landmark examples include glucagon-like peptide-1 (GLP-1) receptor agonists such as semaglutide and liraglutide, which have revolutionized the treatment of type 2 diabetes and obesity by enhancing glycemic control and promoting sustained weight loss. In oncology, cell-penetrating peptides like Tat and tumor-targeting constructs such as peptide–drug conjugates (PDCs) are enabling intracellular delivery of cytotoxins and immune checkpoint modulators with refined precision.
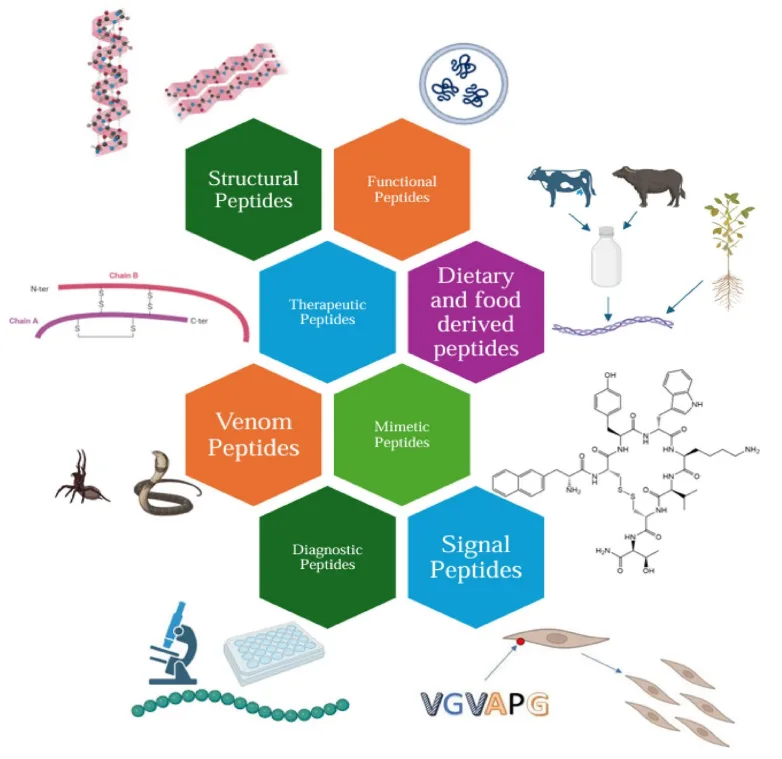
Beyond pharmacology, peptides are gaining ground in dermatological science. Bioactive sequences such as GHK-Cu (a copper-carrying tripeptide) and Matrixyl (a palmitoyl pentapeptide) stimulate collagen production, facilitate wound repair, and reduce signs of aging through targeted dermal signaling. These molecules exemplify the transition of peptides from biological curiosities to clinically relevant, application-driven tools.
As peptide classes diversify—ranging from cyclic peptides and mimetics to venom-derived toxins and food-derived bioactives—they are redefining the therapeutic toolkit with a level of modularity and specificity unmatched by conventional small molecules or biologics.
Engineering Access: Advanced Delivery Systems for Next-Gen Peptides
Despite their therapeutic potential, peptides face significant challenges related to stability, bioavailability, and systemic delivery. Their susceptibility to enzymatic degradation, short plasma half-lives, and poor membrane permeability have historically constrained their clinical utility—particularly for oral or non-invasive routes of administration. However, recent breakthroughs in delivery science are rapidly transforming this landscape.
Injectable routes—including subcutaneous, intravenous, and intramuscular administration—remain standard for many peptide therapeutics due to their capacity to bypass first-pass metabolism and achieve systemic exposure. Yet, these methods can limit patient adherence. In response, researchers are developing alternative delivery platforms such as transdermal patches, inhalable formulations, and nasal sprays that offer less invasive options while preserving pharmacodynamic integrity.
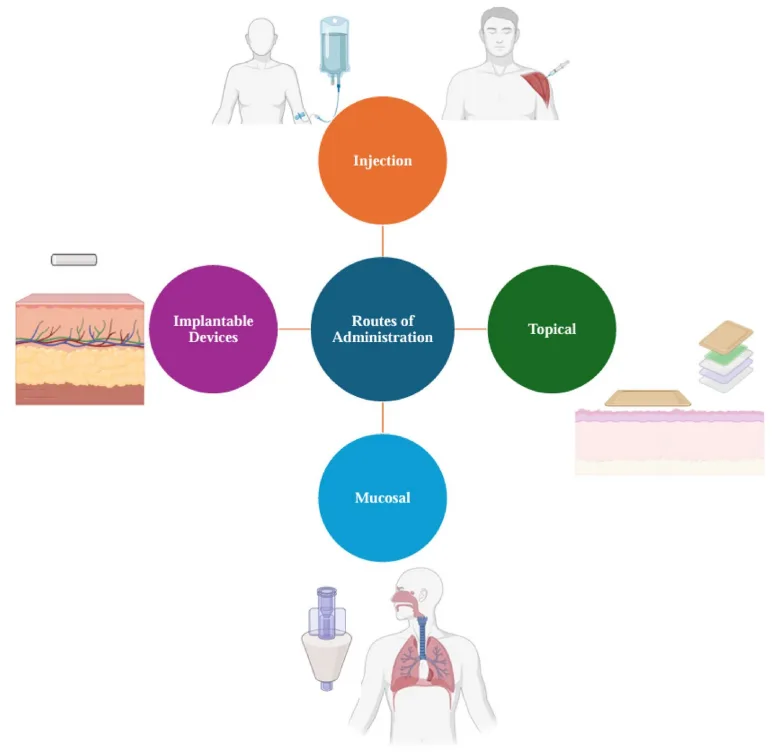
One of the most notable advances is the co-formulation of oral semaglutide with an absorption enhancer (SNAC), which shields the peptide in the gastric environment and facilitates its uptake across the intestinal epithelium—a milestone in oral peptide delivery. Similarly, biodegradable hydrogels, lipid nanoparticles, and dendritic polymers are being employed to encapsulate peptides, protect them from enzymatic attack, and enable targeted or sustained release.
Chemical modifications, such as PEGylation, lipid conjugation, and cyclization, further enhance stability and prolong circulation time. Meanwhile, fusion proteins—like GLP-1 analogs bound to human albumin—extend therapeutic half-life and improve dosing convenience.
Collectively, these innovations not only mitigate historical delivery barriers but also broaden the applicability of peptide therapeutics, making them viable candidates for chronic disease management, immunotherapy, and even regenerative medicine.
Data-Driven Discovery: How AI is Reshaping Peptide Design and Optimization
The integration of artificial intelligence (AI) into peptide science is rapidly reshaping the pace and precision of therapeutic development. Traditional methods of peptide discovery—rooted in combinatorial synthesis and trial-and-error screening—are being eclipsed by data-driven, predictive modeling approaches capable of navigating vast chemical and biological design spaces with exceptional efficiency.
AI enables in silico prediction of peptide structure, activity, and pharmacokinetics, dramatically shortening the discovery timeline. Deep learning models such as AlphaFold have revolutionized protein and peptide structure prediction, offering atomic-level resolution of folding patterns that guide rational design. Tools like PepNN identify peptide binding sites on target proteins, enhancing specificity, while models like AMPScanner and ACPred classify sequences based on antimicrobial or anticancer potential.
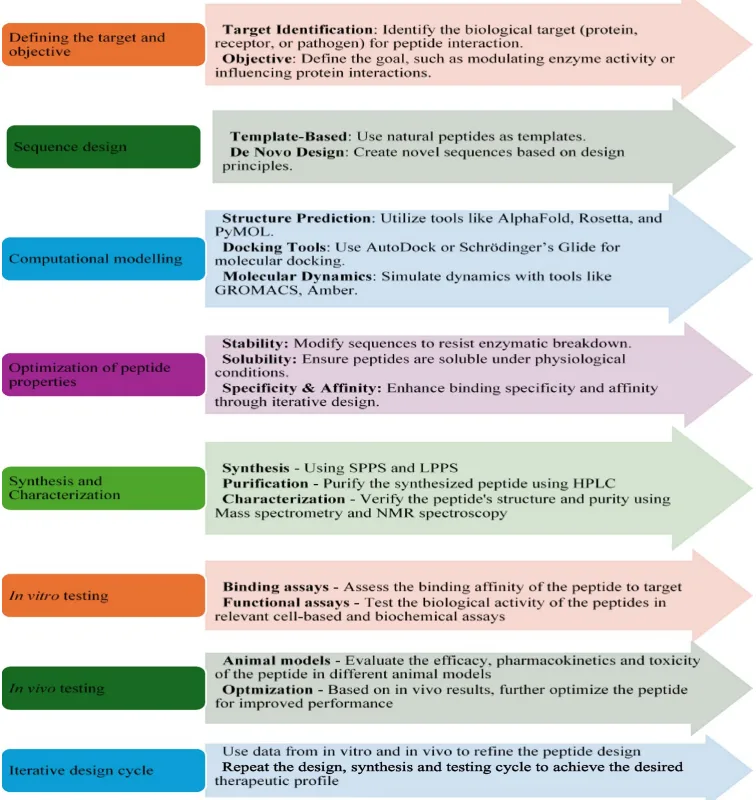
Moreover, generative models, including Variational Autoencoders (VAEs) and Recurrent Neural Networks (RNNs), are being employed for de novo peptide design. These algorithms can generate novel sequences with optimized properties—such as increased half-life, enhanced membrane permeability, or selective receptor targeting—guided by training data from experimentally validated peptides.
AI also plays a pivotal role in predicting peptide–protein interactions, optimizing pharmacodynamic profiles (PK/PD), and even tailoring sequences for personalized medicine based on individual genomic data. These capabilities are particularly relevant in cancer immunotherapy, where neoantigen peptide vaccines and checkpoint-targeting peptides must be adapted to each patient’s tumor mutational landscape.
In essence, AI transforms peptide research from a predominantly experimental discipline into a computationally guided, hypothesis-driven science, unlocking innovations that were previously inaccessible through empirical methods alone.
The Road Ahead: Precision Medicine, Smart Skincare, and the Rise of AI-Powered Peptides
As the intersection of peptide engineering and artificial intelligence continues to evolve, the future of peptide-based applications appears increasingly boundless. Peptides are uniquely poised to drive the next generation of precision therapeutics, where disease-targeted, low-toxicity agents are tailored not only to disease mechanisms but also to individual patient profiles.
In oncology, for example, the development of personalized peptide vaccines and immune checkpoint modulators is redefining cancer immunotherapy—offering highly specific immune activation with reduced systemic burden. Similarly, in neurology, engineered peptides capable of crossing the blood-brain barrier may unlock novel treatments for complex disorders like Alzheimer’s and Parkinson’s disease, where current modalities remain insufficient.
In parallel, the rise of cosmeceuticals—where skincare meets therapeutic science—is being shaped by bioactive peptides designed to stimulate collagen production, modulate pigmentation, and enhance skin regeneration. Supported by AI-based formulation tools and delivery technologies, these innovations are enabling targeted interventions for aging, inflammation, and barrier repair, far beyond conventional cosmetics.
Yet, several challenges remain. Scalability, manufacturing costs, regulatory ambiguity, and the interpretability of AI models are critical hurdles to widespread adoption. Nonetheless, as computational biology, peptide chemistry, and translational medicine continue to converge, peptides are set to become indispensable agents—not just in niche indications, but across the broader landscape of human health, wellness, and disease prevention.
 LinkPeptide
LinkPeptide