Smarter Drug Design: How Artificial Intelligence is Unlocking the Potential of PDCs
Abstract
Peptide–drug conjugates (PDCs) are emerging as powerful tools in precision medicine, designed to deliver potent therapies directly to diseased tissues while sparing healthy cells. Unlike antibody–drug conjugates (ADCs), PDCs use small, flexible peptides as targeting agents, offering advantages such as deeper tissue penetration, lower immunogenicity, and reduced production costs. However, their development has lagged due to limited peptide diversity, unstable linkers, and the lack of predictive evaluation models. Recent advances in artificial intelligence (AI) are transforming this landscape. By enabling smarter peptide design, optimizing linker chemistry, refining payload selection, and enhancing predictive modeling, AI is accelerating PDC research and opening the door to more effective therapies. With the integration of robust databases and advanced computational tools, AI-driven PDCs are poised to become a cornerstone of next-generation targeted treatments, offering new hope for cancer and beyond.
From Magic Bullets to Precision Tools: The Rise of PDCs
In the era of precision medicine, the goal is no longer just to treat disease but to treat it selectively—delivering therapies directly to affected tissues while sparing healthy ones. This is the promise of targeted therapeutics, often described as “magic bullets” that combine efficacy with reduced side effects. Among the most exciting innovations in this space are peptide–drug conjugates (PDCs), a class of medicines that use peptides as guiding molecules to carry potent drugs precisely where they are needed.
PDCs share a conceptual framework with antibody–drug conjugates (ADCs): both couple a therapeutic payload to a targeting unit via a chemical linker. The key difference lies in the targeting agent. Instead of large, complex antibodies, PDCs employ short, flexible peptides. This distinction gives PDCs several advantages—smaller size, deeper tissue penetration, lower risk of immune reactions, and reduced manufacturing costs.
| Drug | Target | Payload | Manufacturer | Indication | Development phase |
| Lutathera | SSTR | 177Lu | Novartis | Neuroendocrine tumors | Market |
| Pluvicto | PSMA | 177Lu | Novartis | Prostate cancer | Market |
| Pepaxto | Aminopeptidases | Melphalan | Oncopeptides | Myeloma | Market |
| CBP-1018 | PSMA;FLORI | MMAE | Coherent Biopharma Suzhou Co.,Ltd. | Solid tumors | Phase I |
| CBX-12 | TOP1 | Exatecan | Cybrexa Therapeutics | Solid tumors | Phase I |
| TH1902 | SORT1 | Docetaxel | Theratechnologies | Breast cancer | Phase I |
| BT5528 | EphA2 | MMAE | Bicycle Therapeutics | Solid tumors | Phase II |
| BT1718 | MT1-MMP | DM1 | Bicycle Therapeutics | Solid tumors | Phase II |
| PEN-221 | SSTR2 | DM1 | Tarveda Therapeutics | Neuroendocrine tumor;Small cell lung cancer | Phase II |
| CBP-1008 | FRα;TRPV6 | MMAE | Coherent Biopharma | Solid tumors | Phase II |
| AEZS-108 | LHRH-R | DOX | Aeterna Zentaris | Endometrial cancer | Phase III |
| BT8009 | Nectin-4 | MMAE | Bicycle Therapeutics | Urothelial cancer | Phase III |
| ANG1005 | LRP1 | PTX | Shenogen Pharma Group;AngioChem | Leptomeningeal carcinomatosis | Phase III |
Despite these strengths, PDCs have developed slowly compared to ADCs. More than 15 ADCs have gained FDA approval, while only three PDCs—Lutathera (2018), Pepaxto (2021, later withdrawn), and Pluvicto (2022)—have reached the market. The reasons are clear: limited peptide diversity, unstable linkers, and the absence of robust predictive tools.
Now, that landscape is beginning to change. With the rise of artificial intelligence (AI), researchers are reimagining how PDCs are designed, optimized, and tested—ushering in a new chapter for targeted drug development.
Inside a PDC: Peptides, Linkers, and Payloads Explained
To understand the promise and limitations of peptide–drug conjugates (PDCs), it helps to look closely at their three essential components: the peptide, the linker, and the payload. Each plays a distinct role, and weaknesses in any one of them can compromise the whole therapy.
Peptides are the targeting engines of PDCs. They fall broadly into two categories. Cell-penetrating peptides (CPPs), such as the well-known HIV-TAT sequence, can slip through cell membranes but often lack selectivity. Cell-targeting peptides (CTPs), like bombesin or RGD analogs, bind to specific receptors on diseased cells, offering higher precision. The challenge? Both types are prone to enzymatic degradation and typically require injections rather than oral delivery. Recent advances—including cyclization, unnatural amino acid incorporation, and AI-driven design—are helping to make them more stable and selective.
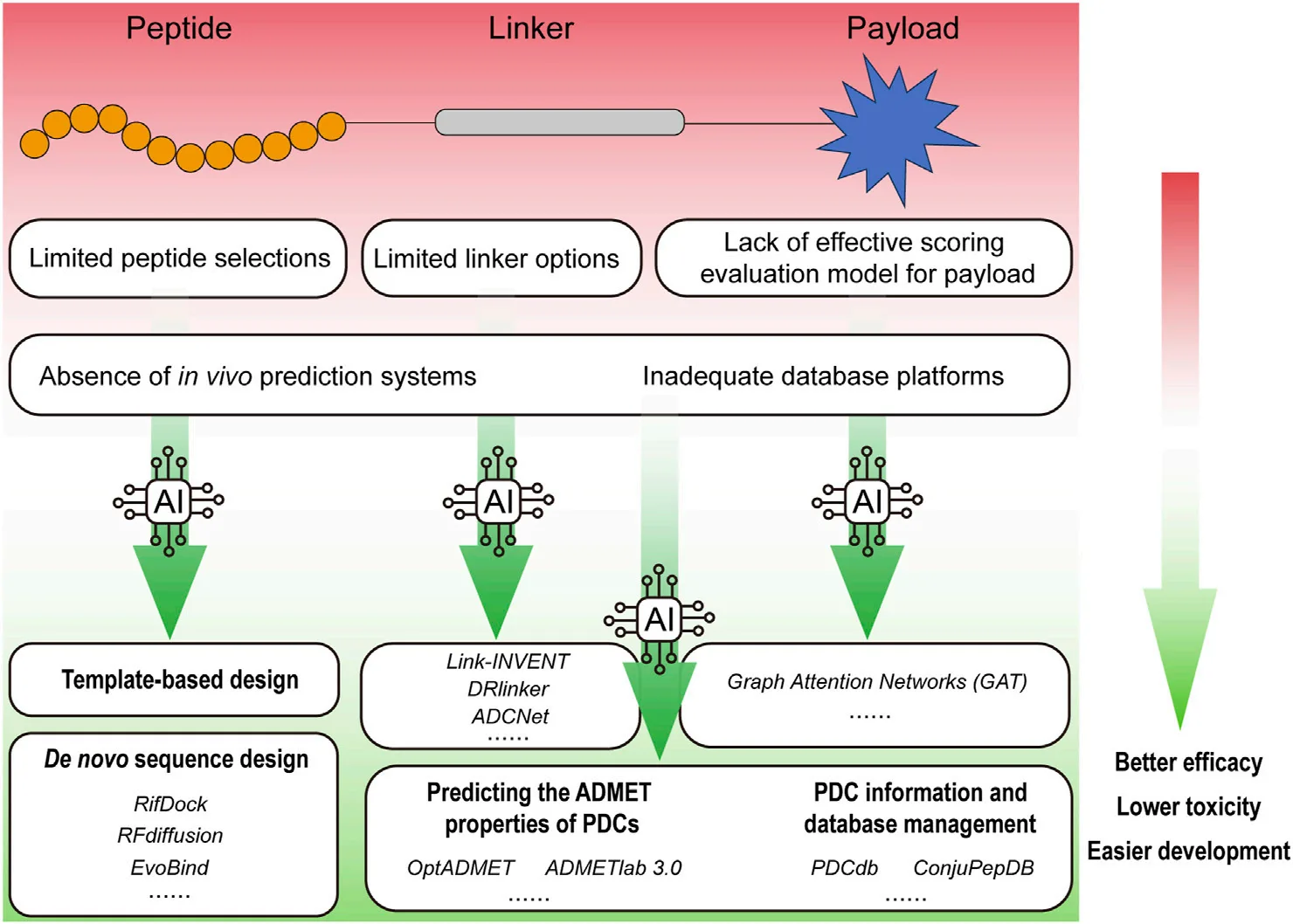
Linkers act as molecular “bridges” between the peptide and the drug payload. They must be stable enough to survive in the bloodstream yet sensitive enough to release the drug once inside the target cell. Cleavable linkers respond to triggers such as acidic pH, enzymes, or redox conditions, while non-cleavable linkers enhance stability but may hinder drug release. The problem is that diversity is scarce: only about 140 linkers are currently used in PDC research, limiting design flexibility.
Finally, the payload is the therapeutic weapon. These range from classic chemotherapeutics like doxorubicin and paclitaxel to radionuclides used for both imaging and treatment. Selecting the right payload is crucial, but traditional approaches rely heavily on trial and error, with no reliable scoring models to guide optimization.
Together, these components define the potential of PDCs—but they also highlight why smarter, more predictive tools are urgently needed.
Why PDCs Lag Behind: The Bottlenecks Slowing Progress
Although peptide–drug conjugates (PDCs) offer exciting advantages over traditional therapies, their progress has been slower than expected. After three decades of research, only a handful have reached the market. The reasons lie in several persistent bottlenecks that continue to challenge researchers.
Limited peptide diversity is one major hurdle. Thousands of natural and synthetic peptides have been studied, yet only a small fraction display the stability, half-life, and receptor specificity required for therapeutic use. Most peptides are quickly degraded by enzymes or cleared too rapidly from circulation, making it difficult to achieve consistent clinical results.
Linker design poses another obstacle. Linkers must strike a delicate balance: they should protect the drug in circulation yet release it efficiently once inside target tissues. Many current linkers either break down prematurely—causing unwanted side effects—or cling too tightly, reducing therapeutic efficacy. With only about 140 linkers reported in the literature, the design space remains surprisingly narrow.
A third bottleneck is the lack of predictive systems. Unlike small molecules, PDCs involve complex interactions: peptide–receptor binding, linker stability, and controlled payload release. Traditional absorption, distribution, metabolism, excretion, and toxicity (ADMET) models cannot capture these dynamics, leaving researchers to rely heavily on costly animal and clinical studies.
Finally, fragmented and incomplete databases slow progress. Without comprehensive resources to integrate peptide, linker, and payload data, rational design becomes guesswork.
These limitations explain why PDCs have lagged behind antibody–drug conjugates (ADCs), despite their theoretical advantages. Overcoming these hurdles requires a new approach—and this is where artificial intelligence is beginning to make a decisive impact.
AI to the Rescue: How Technology is Reimagining PDC Design
Artificial intelligence (AI) is breathing new life into peptide–drug conjugate (PDC) research. By overcoming long-standing bottlenecks in design and evaluation, AI is enabling scientists to move beyond trial-and-error and toward precision-driven discovery. Five areas stand out where AI is making a tangible impact.

1. Smarter peptide design.
Traditionally, identifying the right peptide meant laborious screening or expensive combinatorial libraries. Today, AI platforms like AlphaFold2 and RoseTTAFold can predict peptide–protein interactions with remarkable accuracy, guiding the design of stable and selective sequences. Tools such as RFdiffusion even generate entirely new cyclic peptides with significantly higher tumor affinity than those produced by older methods.
2. Linker optimization.
Choosing the right linker has always been a guessing game. AI-powered tools like Link-INVENT and DRlinker use reinforcement learning to create linkers with optimal length, polarity, and cleavage properties. Meanwhile, ADCNet—initially developed for antibody–drug conjugates—has been adapted to PDCs, allowing rapid in silico screening of thousands of linkers for stability and release efficiency.
3. Payload discovery.
The therapeutic payload must be potent yet safe. AI, particularly graph attention networks (GAT), can now predict how payloads behave inside tumor environments. For example, AI-guided screening has identified exatecan derivatives with seven-fold stronger “bystander killing” effects in drug-resistant cancers.
4. Predictive modeling.
Platforms like OptADMET and ADMETlab 3.0 are improving how researchers forecast absorption, metabolism, toxicity, and distribution. By simulating how PDCs behave in the body, these tools reduce reliance on costly animal models and de-risk early-stage development.
5. Database integration.
Finally, AI thrives on data. New resources such as PDCdb and ConjuPepDB consolidate thousands of entries on peptide sequences, linkers, payloads, and clinical outcomes. When combined with machine learning, these databases accelerate pattern recognition and candidate selection.
Early successes underscore the promise. For instance, MP-0250, an AI-guided PDC targeting VEGF/HGF, achieved a 34% response rate in Phase II lung cancer trials, while AI-optimized somatostatin analogs in Lutathera reduced liver toxicity by 22% post-approval.
Together, these breakthroughs signal a shift: AI is not just a supporting tool but a driving force in the next generation of targeted therapies.
What’s Next: The Future of AI-Driven Peptide–Drug Conjugates
The integration of artificial intelligence into peptide–drug conjugate (PDC) development is more than an incremental improvement—it represents a paradigm shift in how targeted medicines are conceived and tested. By streamlining peptide discovery, fine-tuning linkers, optimizing payloads, and enabling predictive modeling, AI is addressing the very challenges that once limited PDCs to the sidelines of drug development.
Yet, hurdles remain. PDC-specific datasets are still small compared to those for small molecules, making it difficult to train robust AI models without overfitting. Translating AI-designed candidates into clinical success is another challenge: only a fraction of molecules predicted in silico make it to human trials, let alone regulatory approval. Moreover, global health authorities have yet to establish clear frameworks for evaluating AI-driven drug design, leaving companies uncertain about regulatory pathways.
Even so, the direction is clear. As databases expand and algorithms mature, AI will not just accelerate PDC development—it will unlock entirely new therapeutic opportunities. Future PDCs could achieve higher precision, improved safety, and faster time-to-market, offering a powerful complement to antibody–drug conjugates and other targeted therapies.
In short, AI is poised to transform PDCs from a promising concept into a cornerstone of modern precision medicine.
Reference
Zhang, D. E., He, T., Shi, T., Huang, K., & Peng, A. (2025). Trends in the research and development of peptide drug conjugates: artificial intelligence aided design. Frontiers in Pharmacology, 16, 1553853.
https://doi.org/10.3389/fphar.2025.1553853
Bryant, P., & Elofsson, A. (2023). Peptide binder design with inverse folding and protein structure prediction. Communications Chemistry, 6(1), 229.
https://doi.org/10.1038/s42004-023-01029-7
Gayle, S., Aiello, R., Leelatian, N., Beckta, J. M., Bechtold, J., Bourassa, P., … & Paralkar, V. M. (2021). Tumor-selective, antigen-independent delivery of a pH sensitive peptide-topoisomerase inhibitor conjugate suppresses tumor growth without systemic toxicity. NAR cancer, 3(2), zcab021.
https://doi.org/10.1093/narcan/zcab021
Worm, D. J., Els‐Heindl, S., & Beck‐Sickinger, A. G. (2020). Targeting of peptide‐binding receptors on cancer cells with peptide‐drug conjugates. Peptide Science, 112(3), e24171.
 LinkPeptide
LinkPeptide