TXM Peptides: A Promising Antiviral and Anti-Inflammatory Therapeutic Strategy Against SARS-CoV-2
Abstract
The SARS-CoV-2 pandemic continues to pose significant global health challenges, necessitating novel therapeutic approaches beyond vaccines and traditional antivirals. Thioredoxin mimetic (TXM) peptides represent a promising dual-action strategy, targeting both viral entry and inflammatory pathways. These peptides disrupt disulfide bonds within the spike protein receptor-binding domain (RBD), weakening its interaction with ACE2 receptors and preventing viral fusion and syncytia formation. Additionally, TXM peptides exhibit potent antioxidant and anti-inflammatory effects, reducing NF-κB and IRF-mediated cytokine storms. In cell culture and animal models, TXM peptides have demonstrated efficacy in inhibiting viral infection, lowering oxidative stress, and reducing immune overactivation. Their membrane permeability and blood-brain barrier (BBB) crossing further expand their therapeutic potential. As a result, TXM peptides emerge as a versatile antiviral and immunomodulatory platform, warranting further preclinical and clinical investigation for treating SARS-CoV-2, long COVID, and other viral infections.
A New Frontier in Antiviral and Anti-Inflammatory Therapeutics
The COVID-19 pandemic has significantly impacted global health and economies, with SARS-CoV-2 continuing to pose a major threat despite widespread vaccination efforts. Although vaccines have reduced mortality rates, reinfections remain a concern, particularly with the emergence of immune-evasive variants. Moreover, long COVID—a persistent and debilitating condition—affects a significant proportion of individuals even after mild infections. This underscores the urgent need for alternative therapeutic strategies that not only target viral replication but also mitigate the inflammatory and oxidative stress responses associated with severe disease[1].
The SARS-CoV-2 spike (S) protein is a critical target for both vaccines and therapeutics, as it facilitates viral entry into host cells via binding to the angiotensin-converting enzyme 2 (ACE2) receptor. This interaction is heavily dependent on disulfide bonds within the receptor-binding domain (RBD), which stabilize the protein’s structure[2]. Disrupting these disulfide bonds weakens spike-ACE2 binding, thereby hindering viral attachment and entry.
Thioredoxin Mimetic (TXM) peptides offer a promising dual-action antiviral and anti-inflammatory approach. These small peptides mimic thioredoxin 1 (Trx1), a natural oxidoreductase enzyme that regulates redox homeostasis. TXM peptides disrupt the disulfide bonds within the spike RBD, preventing viral fusion and syncytia formation. Additionally, they exhibit strong anti-inflammatory effects, reducing NF-κB and IRF pathway activation, which are key drivers of cytokine storms. Recent research highlights TXM peptides’ potential to lower viral infection rates and inflammatory damage in both cell cultures and animal models, making them an exciting candidate for future antiviral therapies.
How TXM Peptides Disrupt SARS-CoV-2 Infection
SARS-CoV-2 relies on the spike (S) protein to facilitate viral entry into host cells, primarily through its interaction with the angiotensin-converting enzyme 2 (ACE2) receptor. This process is redox-dependent, as the receptor-binding domain (RBD) of the spike protein is stabilized by disulfide bonds, which maintain its conformation for high-affinity binding. Disrupting these disulfide bridges weakens the interaction between spike and ACE2, reducing viral attachment and entry.
Thioredoxin Mimetic (TXM) peptides function by mimicking the catalytic site of thioredoxin 1 (Trx1), a natural oxidoreductase enzyme that regulates redox homeostasis. These peptides contain two cysteine residues, allowing them to efficiently reduce disulfide bonds within the spike protein, shifting them into a less stable thiol form. This conformational disruption hinders the spike-ACE2 binding process, effectively blocking viral entry into the cell[3]. Unlike general antioxidants, TXM peptides target specific cysteine-disulfide interactions, providing a more targeted antiviral effect.
Fig. 1. Disulfide pairs in RBD of SARS-CoV-2 and thiol-reducing antioxidant compounds. (A) Schematic localization of Cys pairs (red) within the RBD and RBM (cyan) of SARS-CoV-2. (B) Chemical structures of AD4/NACA and thioredoxin mimetic (TXM) peptides TXM-CB3, TXM-CB12, TXM-CB13, TXM-CB30, and SD.
Beyond blocking viral entry, TXM peptides also prevent syncytia formation, a pathological feature of SARS-CoV-2 infection where infected cells fuse together, contributing to lung tissue damage. Cell culture studies have demonstrated that TXM peptides significantly reduce SARS-CoV-2 pseudovirus infection, highlighting their potential as a therapeutic agent against multiple viral strains.
Experimental findings further suggest that TXM peptides are more potent than conventional thiol-based agents like N-acetylcysteine (NAC), as they exhibit higher efficacy at lower concentrations. This suggests that TXM peptides could serve as a promising antiviral therapy, particularly against emerging SARS-CoV-2 variants that continue to evolve mechanisms for immune evasion.
The Anti-Inflammatory and Antioxidant Benefits of TXM Peptides
The severity of COVID-19 is closely linked to hyperinflammation and oxidative stress, both of which contribute to tissue damage and immune dysregulation. SARS-CoV-2 infection triggers the excessive release of reactive oxygen species (ROS) and reactive nitrogen species (RNS), disrupting redox homeostasis and exacerbating viral replication. This oxidative imbalance activates inflammatory pathways, particularly NF-κB and IRF signaling, leading to a cytokine storm—a hallmark of severe COVID-19 cases[4, 5].
TXM peptides offer a dual mechanism of protection, acting as both antioxidants and anti-inflammatory agents. By mimicking thioredoxin 1 (Trx1), they neutralize ROS and RNS, restoring the cellular redox balance. Additionally, TXM peptides suppress pro-inflammatory pathways, preventing excessive activation of NF-κB and MAPK signaling, which drive cytokine production. In cell culture studies, TXM peptides have been shown to reduce phosphorylation of ERK1/2 and JNK1/2, key regulators of inflammation.
In animal models, TXM peptides have demonstrated significant anti-inflammatory effects. In a lipopolysaccharide (LPS)-induced inflammation model, TXM peptides lowered cytokine levels, including IL-1β, CXCL10, IFN-γ, and IL-2, all of which contribute to immune overactivation in COVID-19. Furthermore, TXM peptides effectively reduced neutrophil and eosinophil infiltration, limiting tissue damage caused by excessive immune responses.
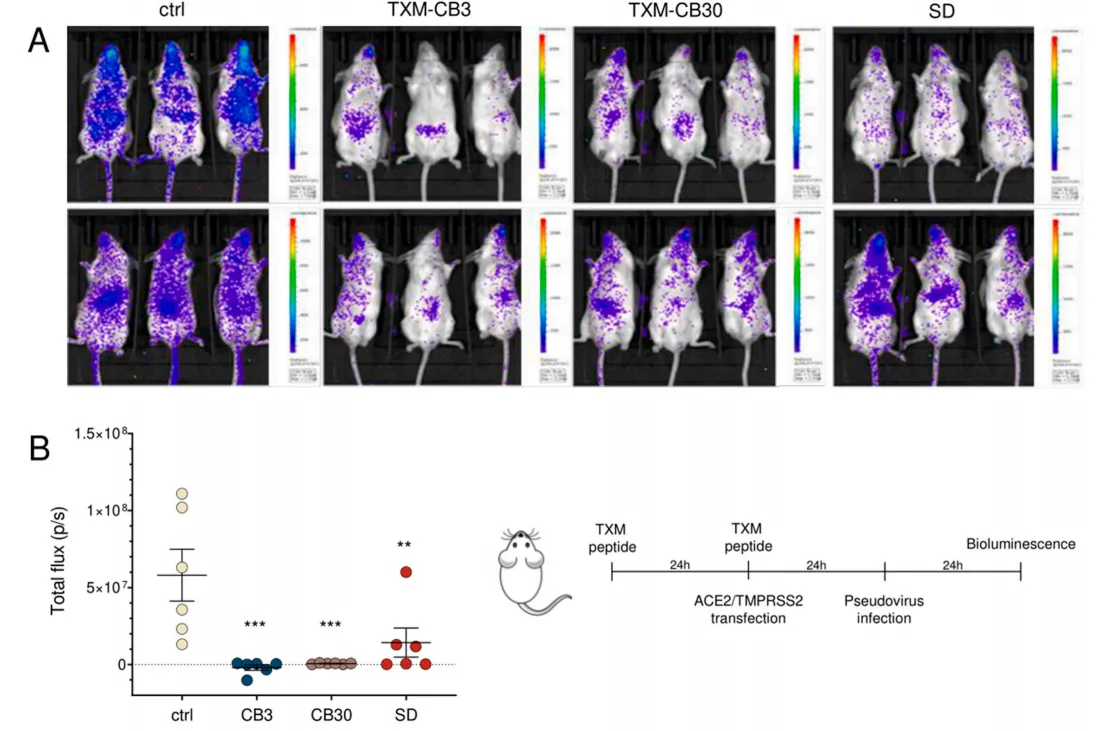
Fig. 2. TXM peptides mitigate pseudovirus infection in vivo. Female 10-week old Balb/c mice were i.p. injected with tested compounds for two consecutive days (2 mg/day). On day 2, mice were intranasally transfected with ACE2 receptor and TMPRSS2 plasmids. Animals were infected with spike protein-pseudotyped viruses, coding for firefly luciferase, on day 3, and 24 h postinfection, bioluminescence was measured (presented here as total flux (photons/s)). Comparison to untreated infected control (ctrl) was analyzed using Brown-Forsythe and Welch ANOVA with Dunnett’s multiple comparison test. ***p < 0.001; **p < 0.01.
Unlike conventional antioxidants, which have limited cellular penetration, TXM peptides are membrane-permeable and exhibit a longer-lasting protective effect. Their ability to cross the blood-brain barrier (BBB) suggests potential applications in mitigating neurological complications of long COVID, making them a promising candidate for future therapeutic development in both infectious and inflammatory diseases.
TXM Peptides as a Drug Development Candidate
The unique mechanism of TXM peptides sets them apart from conventional antiviral and anti-inflammatory treatments, offering several advantages for pharmaceutical development[6]. Their dual-action properties—targeting both viral entry and inflammation—make them a promising candidate for treating SARS-CoV-2 infections and related long-term complications.
1. Broad Antiviral Potential
TXM peptides have been shown to effectively inhibit SARS-CoV-2 infection by disrupting spike-ACE2 interactions and preventing syncytia formation. Importantly, this mechanism is not strain-dependent, suggesting that TXM peptides could remain effective against emerging variants. Additionally, studies indicate that TXM peptides also inhibit HIV pseudovirus infection, implying a broader antiviral spectrum for redox-sensitive viruses.
2. Membrane Permeability and Blood-Brain Barrier (BBB) Crossing
Unlike many antioxidant therapies, TXM peptides exhibit high cellular uptake and can cross the BBB. This makes them particularly valuable for treating neurological complications associated with long COVID and other viral infections. Their ability to modulate oxidative stress and neuroinflammation suggests potential applications in conditions such as viral encephalitis, neurodegenerative disorders, and post-viral fatigue syndromes.
3. Safety and Bioavailability
TXM peptides were found to be non-toxic in vitro, with no observed cytotoxicity at therapeutic concentrations. Their small molecular weight and stability allow them to be rapidly absorbed while maintaining high potency. Compared to traditional antioxidants like N-acetylcysteine (NAC), TXM peptides demonstrate stronger inhibition of viral fusion at significantly lower doses.
4. Potential for Clinical Development
Given their strong in vitro and in vivo efficacy, TXM peptides represent a viable candidate for clinical trials. However, further pharmacokinetic optimization may be necessary to ensure longer retention times in the bloodstream. Future studies should explore:
- Combination therapies with existing antivirals or monoclonal antibodies.
- Optimal dosing regimens for maximum efficacy.
- Expanded applications in treating other viral infections, inflammatory diseases, and oxidative stress-related conditions.
With their unique redox-modulating properties, TXM peptides hold immense promise for antiviral drug development, particularly in combating emerging SARS-CoV-2 variants and post-viral syndromes.
A Novel Strategy in Antiviral and Anti-Inflammatory Therapy
The emergence of new SARS-CoV-2 variants and the persistent threat of long COVID highlight the urgent need for alternative therapeutic strategies beyond conventional vaccines and antivirals. TXM peptides represent a promising breakthrough, offering a dual-action approach that combines antiviral efficacy with anti-inflammatory protection. Their ability to disrupt spike-ACE2 interactions, prevent syncytia formation, and reduce oxidative stress and cytokine production positions them as a potential game-changer in antiviral drug development.
Key Takeaways
- TXM peptides inhibit viral entry by targeting redox-sensitive disulfide bonds in the SARS-CoV-2 spike protein, reducing infectivity across multiple variants.
- They prevent syncytia formation, mitigating tissue damage and disease severity.
- Their potent antioxidant and anti-inflammatory effects help regulate NF-κB and IRF signaling, reducing cytokine storms that drive severe COVID-19 outcomes.
- Unlike many antioxidants, TXM peptides are highly bioavailable, membrane-permeable, and cross the blood-brain barrier (BBB), making them viable for neurological and systemic applications.
Future Research Directions
To fully unlock the therapeutic potential of TXM peptides, further clinical and preclinical studies are needed to address key areas:
- Optimizing pharmacokinetics to enhance bioavailability and retention.
- Evaluating their efficacy against broader viral families beyond SARS-CoV-2 and HIV.
- Testing combination therapies to improve treatment synergy with existing antivirals and monoclonal antibodies.
- Exploring their role in long COVID management, particularly in mitigating neuroinflammation and post-viral fatigue syndromes.
Final Thoughts
TXM peptides stand at the cutting edge of antiviral and anti-inflammatory therapy, providing a targeted, effective, and potentially scalable solution for COVID-19 and future pandemics. Their ability to modulate the redox environment, prevent viral spread, and suppress excessive inflammation sets them apart as a versatile therapeutic platform. As research advances, TXM peptides could reshape the landscape of antiviral drug development, offering new hope for combating viral infections and inflammatory diseases worldwide.
 LinkPeptide
LinkPeptide