Unlocking the Power of Neuropeptide Y: A New Approach to Obesity Treatment Through Thermogenic Fat Regulation
Abstract
Obesity has become a global health crisis, with its associated metabolic diseases presenting significant challenges. Recent research has revealed the crucial role of neuropeptide Y (NPY) in sympathetic neurons, offering new insights into the regulation of fat metabolism and energy expenditure. Unlike its well-known role in appetite regulation in the brain, NPY in sympathetic neurons supports thermogenesis in adipose tissues, particularly brown and white adipose tissue (BAT and WAT). This study shows that NPY sustains mural cells, which are key to thermogenic fat, enhancing the body’s ability to burn calories and prevent obesity. The depletion of NPY in sympathetic neurons disrupts thermogenesis, leading to weight gain without increased food intake. These findings suggest that modulating NPY signaling in peripheral tissues could provide a novel therapeutic strategy for managing obesity. Targeting NPY could shift the focus from appetite control to enhancing metabolic rate and energy expenditure, offering hope for more effective obesity treatments.
Unveiling the Obesity Epidemic and the Potential of Thermogenic Fat
Obesity has reached epidemic proportions globally, becoming one of the most significant public health challenges of our time. The rise in obesity rates has contributed to a surge in metabolic diseases, including type 2 diabetes, cardiovascular diseases, and certain cancers. According to the World Health Organization, obesity is responsible for approximately 2.8 million deaths annually, making it a critical area of concern for both public health systems and researchers. Despite the availability of various interventions—ranging from lifestyle changes to pharmacological treatments—long-term weight management remains an elusive goal for many individuals. While approaches like calorie restriction and exercise can be effective in the short term, they fail to address the underlying mechanisms that drive obesity. A promising area of research involves thermogenic fat, specifically brown adipose tissue (BAT), which has the unique ability to burn calories to generate heat. BAT has garnered attention for its potential to regulate energy expenditure and combat obesity by increasing metabolic rate and fat burning, offering hope for more sustainable obesity treatments.
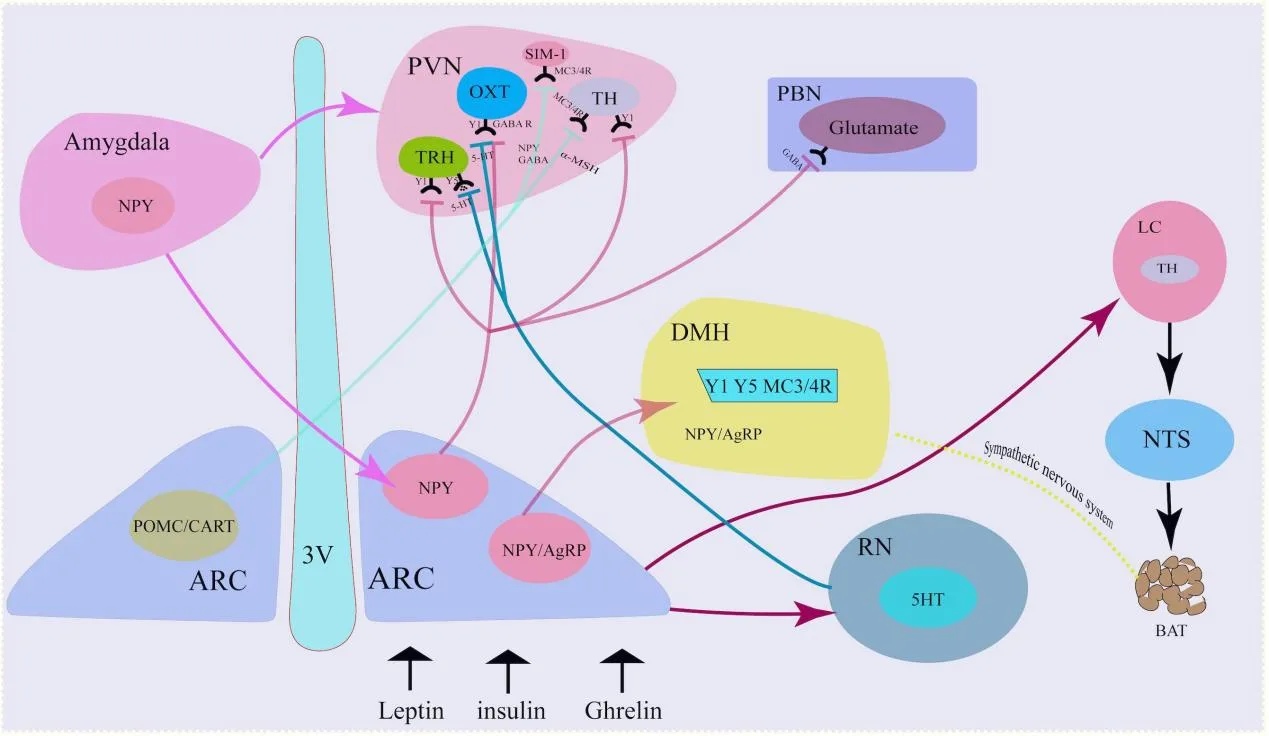
The recent study on neuropeptide Y (NPY) sheds new light on the mechanisms underlying fat metabolism, particularly in how NPY in sympathetic neurons can influence energy expenditure. This research opens new avenues for therapeutic interventions targeting NPY in peripheral tissues to enhance thermogenesis. The study is particularly significant because it introduces NPY’s role in the sympathetic nervous system as a key regulator of fat metabolism, a perspective that is relatively new in the context of obesity treatment. By understanding how NPY sustains thermogenic fat and regulates energy balance, this research could lead to novel strategies for managing obesity without the need for drastic caloric restriction or increased food intake.
Neuropeptide Y: A Hidden Key to Metabolic Regulation in Sympathetic Neurons
Overview of NPY
Neuropeptide Y (NPY) is a potent neuropeptide that has long been recognized for its role in regulating appetite and food intake in the brain. It is one of the most abundant neurotransmitters in the central nervous system and has a well-established role in promoting hunger by acting on specific receptors in the hypothalamus. However, recent findings have expanded our understanding of NPY’s function beyond appetite regulation.
Emerging research has revealed that NPY is also produced by sympathetic neurons, which are part of the autonomic nervous system that controls involuntary body functions like heart rate and digestion. In this peripheral context, NPY appears to play a critical role in regulating fat metabolism, particularly in adipose tissues like brown adipose tissue (BAT) and white adipose tissue (WAT). These discoveries challenge the traditional view of NPY as simply a central regulator of hunger and open new possibilities for its involvement in managing metabolic disorders like obesity.
How NPY Drives Thermogenesis and Fat Metabolism
The newly uncovered role of NPY in sympathetic neurons suggests that it may be crucial for regulating thermogenic fat, which is responsible for burning energy in the form of heat. NPY acts on a specific subset of cells in adipose tissue, primarily mural cells, which are located around blood vessels in both BAT and WAT. These mural cells are essential for the development of thermogenic adipocytes—fat cells that are specialized in burning fat to generate heat. In the presence of NPY, these mural cells proliferate and support the formation of thermogenic fat cells, which in turn enhances energy expenditure. This process is particularly important in combating obesity because it helps maintain a higher metabolic rate, even without an increase in food intake. Through these peripheral actions, NPY sustains the thermogenic capacity of adipose tissues, ultimately helping to regulate body weight by promoting fat burning and energy expenditure. This new role of NPY provides a critical link between the sympathetic nervous system and adipose tissue function, marking a significant step forward in understanding the biological mechanisms of obesity.
Breaking New Ground: How NPY Protects Against Obesity by Enhancing Energy Expenditure
The study exploring NPY’s role in sympathetic neurons utilized animal models, particularly genetically modified mice, to investigate the effects of NPY depletion in peripheral tissues. Researchers removed NPY from sympathetic neurons while leaving it intact in the brain to assess its specific impact on adipose tissue. The results were striking: mice lacking NPY in their sympathetic neurons exhibited a significant increase in body weight, even though their food intake remained unchanged. This finding was pivotal because it demonstrated that the absence of NPY in peripheral neurons could lead to obesity without any direct influence on appetite.
Furthermore, the study observed that this weight gain was associated with a loss of thermogenic function in brown adipose tissue (BAT). BAT, known for its ability to burn calories through thermogenesis, was found to be less active in the absence of NPY. The thermogenic cells in the BAT were less abundant, and their function was severely compromised, leading to a decrease in overall energy expenditure. Additionally, the depletion of mural cells within adipose tissues was observed, which further impaired the thermogenic capabilities of both BAT and white adipose tissue (WAT). This loss of thermogenesis played a key role in the development of obesity in these mice, underscoring the importance of NPY in maintaining the metabolic activity of adipose tissues.
NPY vs. Central Appetite Regulation
An important aspect of the study was the contrast between NPY’s effects on appetite regulation and its role in peripheral fat metabolism. While NPY is well known for stimulating hunger when produced in the brain, the results of this study revealed a different function for NPY in the sympathetic nervous system. In the peripheral tissues, NPY did not affect food intake but instead regulated energy expenditure by enhancing thermogenesis in adipose tissue. This finding is significant because it challenges the long-held view that NPY’s primary role in weight regulation is linked to appetite stimulation. Instead, the study suggests that NPY produced by sympathetic neurons acts more as a regulator of energy expenditure, supporting the body’s ability to burn fat for heat and maintain metabolic balance. This shift in understanding opens new perspectives for obesity treatment, focusing on enhancing NPY’s peripheral effects to regulate weight without relying on appetite control.
Rethinking Obesity Treatment: Targeting NPY for Sustained Fat Burning
The findings of this study suggest that targeting NPY in sympathetic neurons could offer a novel approach to treating obesity, particularly in individuals who are resistant to traditional weight loss strategies. Since NPY produced by sympathetic neurons plays a crucial role in regulating thermogenesis and energy expenditure, modulating its activity could potentially enhance fat burning in adipose tissues. Therapeutic interventions could aim to either boost NPY signaling or mimic its action in peripheral tissues like brown and white adipose tissue, stimulating thermogenic fat without affecting food intake.
This approach would represent a shift away from conventional methods that focus primarily on reducing caloric intake or suppressing appetite. Instead, pharmaceutical treatments could be developed to promote a higher metabolic rate by enhancing the thermogenic function of fat cells. By increasing energy expenditure through the action of NPY, it may be possible to help individuals maintain a healthy weight without the need for extreme dietary restrictions or increased physical activity. This could be especially beneficial for people who struggle with obesity due to metabolic issues, rather than simply overeating.
While the current study provides valuable insights into the role of NPY in fat metabolism, much remains to be explored. Future research should focus on understanding the molecular pathways through which NPY influences mural cells and thermogenesis in adipose tissue. Identifying the specific receptors and signaling pathways involved in this process could lead to the development of more targeted therapies that enhance the effects of NPY on fat metabolism.
In addition, clinical trials will be necessary to assess the effectiveness of NPY-based treatments in humans. Researchers will need to investigate whether manipulating NPY levels in sympathetic neurons can replicate the observed results in animal models, leading to measurable improvements in metabolic health. Furthermore, the potential side effects of such treatments need to be evaluated to ensure their safety for long-term use in obesity management. These future studies will be crucial in translating the promising findings of this research into viable therapies for obesity and related metabolic disorders.
Harnessing the Power of NPY for Future Obesity Therapies
This study offers groundbreaking insights into the role of neuropeptide Y (NPY) in sympathetic neurons and its crucial involvement in regulating fat metabolism, particularly in adipose tissue. The findings show that NPY produced by sympathetic neurons not only affects energy expenditure but also helps sustain thermogenic fat by promoting the proliferation of mural cells in both brown and white adipose tissues. The loss of NPY in these neurons leads to a reduction in thermogenesis, lower energy expenditure, and, ultimately, obesity—without any increase in food intake. These results shift the focus from traditional appetite-regulation mechanisms to a more holistic view of weight regulation that includes the thermogenic properties of adipose tissue.
Given the compelling evidence presented in this study, there is an urgent need for further research to explore the potential therapeutic applications of NPY modulation in obesity treatment. Researchers should focus on unraveling the precise molecular mechanisms through which NPY influences fat metabolism and identifying the best ways to target NPY pathways for therapeutic purposes. Pharmaceutical companies are encouraged to investigate the possibility of developing treatments that can enhance NPY’s action in sympathetic neurons, stimulating thermogenesis in adipose tissue and offering a novel approach to managing obesity. By advancing our understanding of this peripheral role of NPY, we can begin to rethink the strategies we use to combat obesity and its associated metabolic diseases.
 LinkPeptide
LinkPeptide